Uta Mittelstadt BSc, MSc Homeopathic Medicine
Rehana B. Issat BSc Pharmacy, BSc Homeopathic Medicine
Jean E. Duckworth LLB, MSc, MA, RSHom FHEA
ABSTRACT
Objective: To systematically review in the form of a mixed, systematic review, peer-reviewed, randomized, double blind controlled trials and observational studies investigating the effectiveness of the homeopathic treatment for sports injuries. It is sought to identify flaws and weaknesses in the conduction and reporting of trials and to critically analyze issues underlying these implications. This paper seeks to extrapolate a synthesizing argument, and intends to point out in its discussion requirements to overcome potential inconsistencies in future research.
Data sources: Studies were searched from 1990 until December 2012. Electronic databases that are accessible via the UCLan subscription were searched for trials and studies matching the inclusion criteria. Further studies were obtained from personal contacts at the Carstens-stiftung.
Study selection: Studies had to come from peer-reviewed sources and had to have a placebo or a ˈother than placeboˈ control. Treatments had to be homeopathic; either a single remedy or a complex preparation. Pooled-results and systematic reviews were excluded. There were no exclusions by language.
Data synthesis: Trial quality differed extensively. Few similarities and vast differences were noted which complicated a synthesising conclusion. Therefore synthesis was drawn from only four trials that were sufficiently similar in their quality of reporting, conduction and outcome.
Results: Synthesis showed that the homeopathic complex preparation Traumeel, of ingredients in homeopathic mother-tincture and decimal potency, and the single remedy Arnica in X/D and low C potency are effective for sports related injuries, if applied topically following physical exertion or exercise. Homeopathic preparations are very well tolerated and have high patient compliance.
Conclusion: The outcomes reported by the individual trials and studies are inconsistent, and trials have substantial weaknesses. Studies are flawed by wrong application and ignorance of homeopathic principles, by weaknesses of reporting and quality of conduction. We propose that there should be a consensus on the reporting of trials, on a global level, and further postulate the need of a different approach to research when investigating homeopathic interventions.
KEYWORDS:Sports; sports injuries; homeopathy; homoeopathy; randomised controlled trials; observational studies; Arnica; Traumeel, effectiveness
Abbreviations:
CAM – Complementary and alternative medicine
C – Centesimal
D – Decimal
DOMS – Delayed onset muscle soreness
NCH – National Centre of Homeopathy
n.d. – no date
n.p. – no page
NSAID – Non steroidal anti-inflammatory drug
RCT – randomized controlled trial
UKAD – UK Anti Doping
WADA – World Anti Doping Agency
X – English denotation for the D – decimal potency
INTRODUCTION
Background
Homeopathy was developed by Christian Friedrich Samuel Hahnemann (1755 – 1843) in response to the frequently invasive treatments that patients were subjected to by the orthodox treatment practices of his time (1-3). He recognized the healing potential that lay within a substance once it was ultra highly diluted, beyond the existence of molecules of active ingredient, and rhythmically shaken at various stages of the dilution process. He researched into ways for best administering and prescribing this potent, yet gentle alternative approach to patient health care (4, 5).
The principal tenets of this ˈnewˈ treatment are its holistic nature, taking into account the collective of all symptoms that the patient expresses; the law of similar, treating patients with a medicine that produces similar symptoms of illness in a healthy individual; as well as the prescription of a remedy selected to match the patientsˈ unique presenting state of health, the so called individualization (5). Such proceeding is much in opposition to standard conventional practice and even more so irritated, not to say affronted, medical thinking at the time it was first put into practice by Hahnemann (1-3).
Homeopathy remains up until today heavily criticized for the absence of a plausible explanation for its action mechanism, as no investigation has yet been able to ascertain its modus operandi. All the same, there is evidence of efficacy of homeopathic treatment from research (6), and there are extensive experiential accounts from patients reporting of successful treatment practice. Latest affirmation of its effectiveness is reported, from a domain that normally is thought to be rather conventionally oriented; from professional sports.
In 2012, Kisser and Bauer (7) published a report on the incidence of sports injuries in the lay and professional, physically active population of the European Union, from 2006 to 2008. Sports injuries, at the time, made up 14% of all injuries requiring medical care. The authors reported that, statistically, most injuries in team-sports occurred in football. Football in all age groups up until the age of 60 years, according to the authors’ findings, was accountable for over 70% of injuries contracted during sports (7). According to Zittlau (8), in Germany in 2008, these figures were comparable to data appraised of professional football only.
In the professional sector, tournament schedules and league timetables determine when an athlete should have attained highest levels of fitness, and should be able to deliver his maximum performance (9). The industry behind the professional players sees a monetary deficit if the athlete is unable to be out in the playing field (7).
The continuous intensive work-out, on-demand peak activity in games, and the only minimally conceded time for recuperation between competitions, matches and return games, over stress the athletes’ physique (8, 10). Increasingly therefore, there is demand for a form of treatment that in turn is not stressing the body in the process of recovery from injury or exertion. As such the alternative therapies are continually gaining ground in professional sports (8, 11).
In 2008, a survey first brought light into the extensive popularity of homeopathy in professional football in Germany (11). In the first and second Bundesliga, the professional German football leagues, 92% of clubs provided their teams with homeopathic treatment; 75% of these clubs used complex therapeutic prescribing, the remainder also opted for single homeopathic remedies (11).
The medical teams of the Bundesliga clubs see the benefit of homeopathy, in the absence of side-effects and in the lack of restrictions by the official doping associations (8,10,11); and also appreciate that homeopathic remedies are not known to interact with conventional or other therapies and can therefore easily be combined with other interventions (12). This latter fact makes homeopathic remedies valuable, in a quest to enhance conventional interventions (13).
Sports persons evermore demand to be treated by this gentle, rapid and effective treatment approach that homeopathy offers (14), and have recognized the inherent potential to augment resilience and accelerate recovery (15). There are even experiential reports of the capacity of homeopathic remedies to improve performance (10; 16). Homeopathy therefore offers a treatment that is predestinated for the active sports-person, and it speaks for itself that in professional sports such a vast number of clubs of one of the major football leagues of Europe, resort to homeopathy for the treatment of their highly trained athletes.
Literature review
There is some evidence from individual trials and studies of the effectiveness of homeopathic prescribing for injuries that can be contracted during physical exertion related to sports activities. Yet, the outcomes reported are inconsistent. No studies or systematic reviews have principally focused on the generalized description of sports injuries in their appraisals. Solely one systematic review was found that investigated the effectiveness of homeopathic remedies for delayed onset muscle soreness, an injury that may be contracted during sports related exercise. The authors of this review came to the conclusion that the investigated homeopathic remedies were not effective beyond placebo for DOMS (17). Another systematic review investigating the efficacy of Arnica (18), a remedy widely recognized for its applicability to injuries relating to sports, such as symptoms of trauma and muscle soreness (19), did not substantiate the claim of efficacy of this homeopathic prescription.
Our paper therefore seeks to critically analyze the available research from peer-reviewed, published, randomized controlled trials and observational studies in the form of a mixed systematic review, with the aim of extrapolating a synthesizing argument, and identifying possible reasons for the potential inconsistency of the results reported. The aim is also to draw attention to the probable causes of flaws and weaknesses in the conduction and reporting of these trials, with the broader purpose of informing future research into this topic and homeopathy at large.
METHOD
Data Sources
Relevant databases available via the UCLan subscription were searched for trials and studies that met inclusion criteria defined for this review. We inspected the findings from the following databases: Academic Search Complete, AMED, BioMed Central, BMJ Journals Online, CINHAL Plus with Full text, Cochrane Library, EBSCO host EJS, Embase, Medline (with Full text), OVID (Journals and eBooks), Pro Quest, ScienceDirect, SPORTDiscus Full text, Springer Link, Wiley Online Library.
It was found that there is no consensus on the spelling of the term “Homeopathy” in most of the electronic databases, and searches produced different results for each spelling of the term used. Therefore the search terms we used were: ˈHomeopathyˈ, ˈHomoeopathyˈ, and ˈSportsˈ and ˈSports injuriesˈ. A Boolean search was performed using ˈANDˈ as well as ˈORˈ to combine the above terms. There were no exclusions by language of the published trials or studies. Because we anticipated that the status of databases described as international, could provide access to research published in languages other than English, this search was also conducted with respective translations of the search terms in German, French, Spanish and Portuguese. We found though, that little is published in languages other than English and rarely is available full-text in the original language. While this is probably due to restrictions in the UCLan subscription, 2 such foreign language primary studies that met the selection criteria were included in this review. These had been acquired for a previous work of the primary researcher of this review, and at the time had been obtained through personal contacts at the Carstens-stiftung, Germany. They were translated from their original language to English.
Studies selected from journals had to have been published within the time-range 1990 to December 2012. It was emphasised to select only primary studies and not use repeat publications. Meta-analyses or systematic reviews were not included in our elaboration but we did search these for references of primary studies relevant to our review topic. As our focus was on trials on human subjects, animal studies were excluded. Trials available as title or abstract only, where it was not possible to retrieve the full-text study via other sources or personal contacts, were also excluded; as the applicability to our research question could not be verified.
The studies and trials had to have been published in peer-reviewed journals. Peer-review denotes, that prior to their publication trials have been critically appraised by experts in the topic investigated by the study (20). Non-peer-reviewed publications were not included as we considered peer-review an important quality criterion we intended to be expressed in this review. Therefore, this status was ascertained by selection of the specific criteria in our database search. Where this was not possible it was verified via identification of the term ˈpeer-reviewˈ as mentioned on the websites of the journals where the studies had been published. If the websites did not explicitly mention this, personal correspondence with the journal editors was accepted as certification of this criterion.
Studies were included if they provided a control. This could be a placebo or a ˈother than placeboˈ control. Placebo as the control treatment describes an ineffective medicine; a medication that is identical to the active test substance but lacking the active ingredient (21). A ˈother than placeboˈ control could be any other medicinal substance of active ingredient (22).
For inclusion, trials had to have been conducted in a randomised manner. Randomisation describes the allocation of participants of research investigations to either a treatment or control group. This allocation is concealed such that the assignment to either group is by chance and unknown to the participants (23). The inclusion criterion of using RCTˈs in this review assures randomization is met for this trial method. The observational studies lack this randomization. By nature of their design and methodology the same form of randomization is impossible (24). Therefore, these studies were exempt of this randomisation as an inclusion criterion.
The trials and studies could follow the classical homeopathic approach, as Hahnemann had developed it (5), or could explore the approach of complex homeopathic prescribing. Complex homeopathy describes an approach whereby a group of single homeopathic remedies are given in a formulation, matching the more general spectrum of a disease as expressed by numerous patients (25).
Trials could be non-individualised or individualised. Individualization is a principle dictum fundamental to homeopathy (5). This refers to the selection mode of the remedy. The homeopathic remedy given to the patient is selected according to the idiosyncratic symptom descriptions of the patient (22). In the context of this review, this pertains to the selection mode of the homeopathic remedy for each participant. It as such would presuppose that each participant would have been interviewed individually for his or her idiosyncratic symptom expressions, and a remedy consequently would have been selected to match each participant case specifically. Non-individualized thus refers to a generalized mode of prescribing whereby all participants of a trial receive the same homeopathic medicine, ignorant of their individual disposition and nature that is normally respected in, and fundamental to, a homeopathic case-taking (22).
Study Selection
We ended up with 11 studies that met the inclusion criteria. 8 were randomized controlled trials, one of which was a pilot to an RCT, and 3 were observational studies. Two studies were in languages other than English, namely, German and Norwegian. Both came from prior contact to the Carstens stiftung. They had been available as title, respectively abstract only in our database search via the UCLan subscription. Both were translated into English for our appraisal.
One study was included that had been undertaken in a cross-over design (26). This trial design alternates the participants of the respective remedy and placebo group, producing 2 trials with the same participants. Participants are allocated to both groups, one after the other, as such the participant number is considered to have been doubled (27). With this design, randomization is overcome and is as inclusion criterion negligible. Thus we considered the inclusion of this study to be acceptable.
One study, namely that of Schmidt, C. (28) published in the Journal of the American Institute of Homeopathy, was included in this review albeit a newest appraisal by Mathie et al.(22) categorizing it as a non-peer-reviewed publication. This study had been referenced as a publication in the ˈJournal of the American Institute of Homeopathyˈ. The American Institute of Homeopathy today publishes a Journal by the name ˈThe American Journal of Homeopathic Medicineˈ. On its website the American Institute of Homeopathy describes this Journal as a peer-reviewed source. As we were uncertain, due to the different nomenclature of the name of the Journal cited as publishing source of this article, we established personal contact for further clarification. It was thereby verified that the trial by Schmidt, C. (28) is a publication of the American Institute of Homeopathy, and as such has been subjected to peer-review prior to publication (Personal communication 2013 February 06). Consequently we saw no reason to consider the inclusion criteria for this study as not met.
We also included 2 studies that did not explicitly state sports injuries or sports-related physical damage in their title, but used more generalized terms such as ˈinjuriesˈ (29) and ˈmechanical-injuriesˈ (28). Here criteria for inclusion were met via the description and definition of the generalized terminology used. Schneider et al. (29) in their abstract and introduction specified that they investigated a trial population that was suffering of musculoskeletal injuries. The authors further went on to distinguish those to be “sprains, strains, contusions etc., of the ankles, knees and hands” (p.22). These injuries describe an array of lesions or physical harm that can be considered to belong to the symptom complex defined as sports injuries (30).
Similarly, Schmidt (28) defines mechanical injuries to be principally “injuries from exertion, sprains, contusions, bruises” (p.186) and describes the inducing factor of such injury on the participants of her trial, to originate from a “foot-race” (p.186).
A sports-injury is considered to be an injury contracted during exercise and physical exertion, as they are commonly undertaken in sports and sports related activities (30). Usually this incorporates injuries that affect “muscles, bones, tendons, cartilage and associated tissues” as well as “sprains, strains, fractures and contusions” (30 p.8). The trials included in this review investigate delayed onset muscle soreness (DOMS), a hurt and tenderness, with sensitivity to touch, that appears following physical activity and is characterised by belated set in of symptoms, usually 1 to 2 days after the physical exertion. This soreness can last up to two days (30, 31). There is also appraisal of acute discomfort from over-exertion, which is described as “stress and physical trauma” by training (30 p.20), and there is analysis of musculoskeletal trauma, the impact “on organic tissues of a mechanical action, extrinsic or intrinsic; single or repeated” contracted during sports (32). One study focuses on epicondylitis, an “overuse injury in the adult elbow” (30 p.110) and another on tendinopathy, a generalized term for injuries of the tendons, of overuse, or inflammatory processes (33, 34). An overview of the studies included in this review is given in Table 1 below.
Data extraction and assessment of trial quality
This review was not aimed at providing quantitative and numeric data, extracted and appraised from the included trials and studies. We considered this an inappropriate evaluation for this review. The included studies investigated similar, but differing indications and conventional diagnoses that were treated with different homeopathic remedies, which in turn were prescribed in different potencies. Such fundamental variance cannot permit a generalised appraisal (35, 36), not in treatment, nor in research. Therefore we considered this argument applicable to the conduction of this mixed-systematic review, and find it controversial to quantitatively review and compare investigations of these homeopathic treatments. For a qualitative appraisal though, this argumentation is not valid, as the same difficulties arise for any investigation trialling homeopathic treatment interventions indifferent of the characteristics of the individual studies.
The quality of studies was appraised by verification of the extent of reporting of study relevant details as postulated by the respective CONSORT statements for RCT (37), the adjuvant document for homeopathic interventions, the REDHOT guidelines (38) and the STROBE document with guidelines for observational studies (39).
The outcomes presented by the individual trials were compared for their reports of effectiveness. We appraised the trials and studies for their possible biases, flaws, limits and the trial methodology used for investigation. The research model used to conduct the studies, was furthermore critically explored for its applicability to a homeopathic trial context.
We sought to perform a synthesis of our findings, of the reported outcomes and the quality of conduction and reporting of the randomized controlled trials and the observational studies. In order to maintain in accord with the demands of the characteristics considered fundamental to a mixed systematic review, we appraised the differences and resemblances of the studies investigated in this review (40).

RESULTS
Characteristics of included trials and studies
All studies used generalized homeopathic prescribing that against the homeopathic dictum of individualization (5), administers one prescription for all, indifferent of prior idiosyncratic appraisal of the individual and his disease.
Seven of the selected RCTs investigated delayed onset muscle soreness (DOMS) and related parameters (26, 41 – 46). While Tveiten et al. (43), Tveiten et al. (44) and Vickers et al. (46) only used the terminology ˈsorenessˈ it became evident from the description of their trial procedures that they too investigated DOMS and not another form of discomfort that could be experienced as soreness, such as the discomforts associated with over-training (47). These are of a more acute onset than DOMS, and are colloquially frequently wrongly described as muscle soreness (47).
Schmidt (28) in the title of her study indicated she was investigating the indication of “subcutaneous mechanical injuries” (p.186). In the abstract she reported “acute muscle injury…from over-exertion” (p.186), and in her methods section declared that participants had to report “muscle soreness attributable to the race” or, had to have an anticipation of this (p.189). This caused some confusion, as she had asked her participants to apply the ointment “immediately [after the race] to the most sore areas” (p.190). In an anticipated soreness this would only have been possible at a later stage, upon expression of symptoms. Only after the occurrence of symptoms therefore, would the participants have been able to identify the most affected areas. As such, an anticipation of muscle soreness, in our opinion, would have fallen into the treatment category of delayed onset muscle soreness. Hence the author was imprecise in her inclusion criteria. As she furthermore did not clarify on the number of participants that experienced soreness at a later stage, and as participants had to report back, or were contacted in a time span of 24 to 72 hours, it cannot be identified if participants actually reported of the instantaneous soreness after the race, or of one that possibly set-in sometime after the race. Despite this weakness in her inclusion criteria, we acknowledged the intention of the author to explore “acute” expressions (28 p.186), as she did not explicitly mention delayed onset of muscle soreness in the remaining context of her research. Therefore, in keeping with the above definitions, we listed the trial by Schmidt (28), in our review, as investigating symptoms from over-straining.
Epicondylitis was the indication under investigation by Birnesser, Oberbaum, Klein, and Weiser (48), and the effect on tendinopathy was explored by Schneider, Klein, Stolt, and Oberbaum (49). Musculoskeletal trauma as from sprains, strains and contusions were the injuries at the centre of the investigations by Schneider et al. (29).
The scales and parameters used to investigate trial outcomes differed in the individual studies. Main scales used were the visual analogue scale (26, 28, 43, 44, 46), and the Likert scale (41, 44, 46). Other scales and questionnaires were used to rate parameters investigating restitution time, stiffness, pain variables, symptom free days, functional impairment, range of motion, swelling, maximum muscle torque, and strength.
The control of the eight randomized controlled trials was in all studies placebo. Of the observational studies, all three compared the homeopathic complex preparation Traumeel versus a ˈother-than-placeboˈ control, that is, versus an active medication (22), in these cases a conventional treatment, namely non steroidal anti-inflammatory drugs (NSAID) (48); such as Diclofenac (49), and other not further specified conventional medicines (29). This is visualised in Table 1 above.
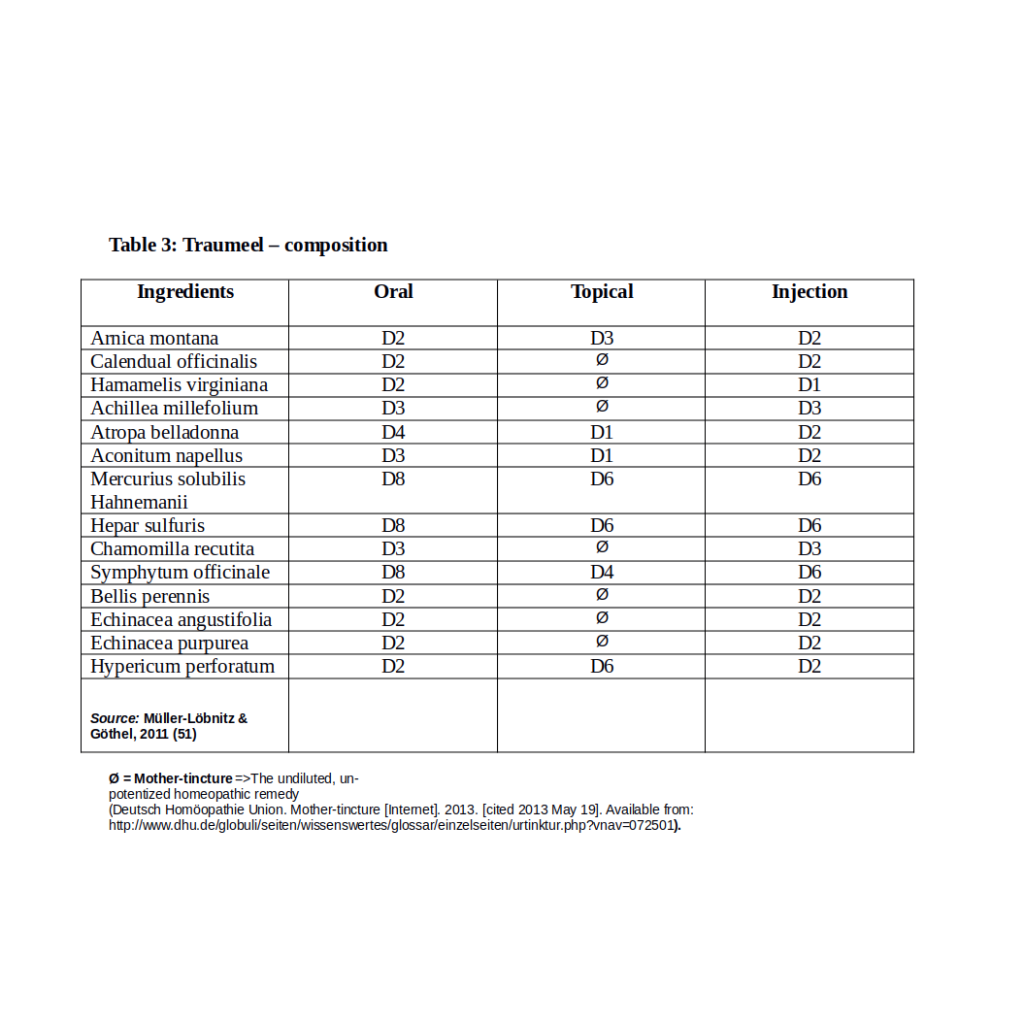
Arnica (18) and Traumeel (50) are the best investigated homeopathic remedies, and in this review these are also the most represented. Six trials investigate Arnica used as a single preparation, two use Arnica in a complex compound that is, as a preparation containing two or more remedies (25), and three studies are on Traumeel a complex compound containing Arnica (Table 2). The potencies of homeopathic preparations used in these trials vary greatly. Arnica as a single preparation was used in potencies X1(D1), X4(D4), X30(D30), C6, and C200 and was used in a C30 in combination with Rhus toxicodendron, and Rhus toxicodendron and Sarcolactic acid, respectively, in two trials using complex preparations. In the trials using Traumeel as treatment compound, comprised of 14 different homeopathic remedies, Arnica is contained in low X/D potencies. The potency of Arnica and the other ingredient homeopathic remedies, included in this complex formulation, differs depending on the route of administration of the preparation. In oral intake and administration via injection, Arnica is used in X/D2, and as a topical agent Traumeel contains Arnica in X/D3 (51). While the oral and injection complex contain only potentized homeopathic remedies, the topical formulation contains homeopathic substances of potentized form and un-potentized, undiluted mother-tincture. These compositions of Traumeel are indicated in Table 3.
It could not be determined whether there was a difference in composition of ˈTraumeelˈ as reported by Schneider et al. (29), and the preparation ˈTraumeel Sˈ, as it was described in Birnesser et al. (48) and Schneider et al. (49). The manufacturer on his website solely differentiates one other Traumeel product range, specifically described as destined for use in the treatment of animals, and largely denoted by the letters T and LT (52). Within the context of this review, we therefore considered the products ˈTraumeelˈ and ˈTraumeel Sˈ, as used in the above studies, to be identical.

The route of administration, of remedies or control in these studies, was in 7 trials oral, in 2 studies topical, in 1 trial injection, and in one appraisal topical or topical and oral in combination (Table 2).
Sample size ranged from just 20 participants (42) to 400 (46) (Table 1). Whilst most RCTˈs conducted their trials on a sample population participating in footraces or marathons (28, 43, 44, 46); Jawara et al. (41) and Vickers et al. (45) trialled their participants after short term bench stepping exercises, Plezbert and Burke (42) and Raschka and Trostel (26) used other short term exercises to induce muscle soreness. The trials on Traumeel were conducted with participants that had been conventionally diagnosed as having tendinopathy (49), epicondylitis (48) or other sports related acute injuries (29).
Tveiten et al. (43) used only male participants in their trial. Tveiten et al. (44) did not mention whether their sample was male, female or both. All the remaining trials were conducted using mixed, male and female, samples.
The degree of sportive experience or regularly conducted practice of physical exercise of participants varied from trial to trial. Vickers et al. (46), Tveiten et al. (43) and Tveiten et al. (44) reported using runners at marathons as population sample. Vickers et al. (46) mentioned that they included participants from races of different lengths, ranging from 2 to 50 miles, and as such possibly also included participants conducting their very first race. Tveiten et al. (43) and Tveiten et al. (44) included participants of the 42km Oslo marathon that by length of distance is unlikely to be run by first time runners. Their participants were reported to conduct regular exercise encompassing a high number of kilometres per week, certainly a distance beyond that of amateur or occasional sports persons. We therefore considered their participants as professional runners. Jawara et al. (41) indicated their sample to have been a relatively even spread of amateur and regularly exercising participants. They, Plezbert and Burke (42) and Vickers et al. (45) excluded participants if they conducted certain strenuous sportive routines on a regular basis. Raschka and Trostel (26) mentioned no exclusion criteria or prior to trial interrogation of participants for their levels of habitual physical exertion. Schmidt (28) considered the sample population to be principally amateur runners as her trial was conducted at company employee races. She further stated that no screening for customary sportive engagement of participants had been conducted.
The duration of trials varied from 2 days (28) to 3 months for follow up time on Traumeel versus conventional treatment (29). Five RCTˈs were undertaken over the period of 5 days (42, 43, 44, 45, 46). Jawara et al. (41) extracted data for 7 days, Raschka and Trostel (26) for 4 days. The observational studies generally were of longer duration with Schneider et al. (29) as mentioned the longest, Schneider et al. (49) continuing for 28 days and Birnesser et al. (48) lasting for 2 weeks.
In the majority of RCTˈs the study participants were instructed to begin taking the remedy or respectively the placebo on the day prior to the sportive event or the trial exercise regimen (26, 41, 43 – 46). Only Schmidt (28) and Plezbert and Burke (42) administered the trial substances after the race. The observational studies began treatment following diagnosis (29, 48, 49).
Outcomes reported and flaws noted by authors
A slight positive effect of Arnica on soreness was reported by Tveiten et al. (43) yet it was not of statistical significance. Authors assumed that the small sample size of just 36 remaining participants was the main flaw of their trial. They also pointed out that the possibility of their Arnica group being “in better shape” (p.3631) could be accountable for this. They noted that coincidentally, their Arnica group was, at baseline, accustomed to more exercise per week than their placebo group. Blood parameters they investigated were never of a statistically significant difference, and restitution time was found to be unaffected by Arnica.
Schmidt (28) came to the conclusion, following the trial of topical Arnica ointments of potency X1 and C6 versus placebo, that results clearly favoured Arnica. Patients experienced “considerably more relief from Arnica than from placebo applied topically to acute muscle soreness from over-exertion” (p.192) following a foot race. She only took one measurement from the participants for her results. Schmidt (28) pointed out that a flaw to her trial probably was the choice of placebo. She noted that petroleum jelly was considered to have an inherent calming effect that could be experienced as a relief from muscle soreness. Therefore she expected to have some deviation in measurements from the placebo group. The author also pointed out that the subjective responses given by participants could be a source of bias as the individual interpretations of pain and relief could be rated positively or negatively depending on the individual and his or her general composure and could therefore impact results. She also indicated that maintaining contact with participants was difficult and described it as “the most frequent obstacle” to obtaining responses and maintaining a higher participant number (p.191).
Jawara et al. (41) found their results to “clearly favour homeopathy” (p.14) but maintained that results showed clinically but not statistically significant relief of soreness, while reporting shortened time until symptom elimination. They also found that their groups differed at baseline, as participants who conducted habitual physical exercise experienced less soreness than participants for whom regular exercise was not customary. They used bench stepping exercise to induce muscle soreness. They further stated that their investigation was not sufficiently powered and future research would require a much larger number of subjects. They too, as did Schmidt (28) in her trial, note that interpretation by participants could be a source of flaw for certain outcome measures.
In 1997, Vickers et al. (45) concluded in their trial investigating homeopathy for delayed onset muscle soreness, that there was no benefit of the homeopathic compound preparation of Arnica, Rhus toxicodendron and Sarcolactic acid in potency C30. They elaborated that their exercise regimen chosen to induce DOMS, bench stepping, may have been inappropriately chosen. They found recruitment of suitable participants impaired by this type of exercise. Therefore their sample size was small with just 57 participants. There was great variation in the extent of soreness described by participants and the extent of DOMS experienced was influenced by prior customary exercise undertaken by the individual subjects. Of the reported side-effects, the majority occurred in the placebo group. One report of side-effects by a participant of the homeopathic group was considered by the authors to be disassociated to the trial, while 3 other reports from this group had similarity in the symptoms they reported.
In a second trial by Tveiten et al. in 1998 (44), the authors presented in their research results, that muscle soreness and running time were improved by Arnica D30 following a marathon race. Like in 1991, blood parameters were inexpressive following the intake of homeopathic Arnica, and the time of restitution was also unaffected. In this trial participant numbers were also low and none of the measured outcomes provided statistical significance.
Vickers et al. (46) undertook another trial, this time with 400 marathon runners. They had sought to replicate the two prior studies by Tveiten et al. (43, 44). Their findings though did not confirm that homeopathic Arnica X/D30 had the capacity to decrease DOMS. As they recruited participants from 114 different races of different duration, the researchers did not take measures directly after the race. They did not consider this to have been a decisive flaw to their trial, as the impact of a curative agent for DOMS should not only be effective directly following a race, but should last longer. Adverse effects reported were distributed evenly between groups and were therefore believed not to be related to remedy or control medication. Vickers at al. (46) considered the positive results of the Tveiten et al. (43, 44) trials to be down to “beta-errors resulting from multiple testing” (n.p.).
In Plezbert and Burke (42) no outcomes confirmed a clinical efficacy of Arnica in C200 on DOMS or other symptoms pertaining to muscular disorders originating from exercise. The authors rated their chosen regimen for induction of DOMS as appropriate and used the participants’ non-dominant arm for the exercise. Blood samples showed no difference from remedy to placebo group. The researchers’ stressed that future investigations into homeopathy should not be ignorant of the fundamental dictum of individualization, and should appraise individual symptom pictures of participants, in order to identify an individualized prescription for each participant.
The conclusion of Raschka and Trostel (26) stated that Arnica D4 had no improving influence on muscle soreness. Raschka and Trostel (26) investigated blood parameters, pain measures and muscle strength. In their trial too, blood measures were unremarkable. They used concentric weight training to induce muscle soreness. They mentioned that with their selected study methodology the generation of a positive outcome was not possible; drawing attention to the design they were using. The authors used the cross-over design that alternates participants between the treatment and placebo group. Albeit the fact that due to this alternation, sample size could be considered to be doubled, the authors mentioned the small participant number to be a flaw to their trial.
Of the observational investigations into the non-inferiority of Traumeel, Birnesser et al. (48) reported “significantly improved scores” (p.199) on all investigated variables, except for the time of action onset. Further did they stress that the injection of Traumeel S compared to NSAIDˈs was equivalent, even superior in some measures. Participantsˈ judgement of treatment, compliance and tolerability was considered higher for Traumeel than for NSAIDˈs. The researchers noted that participants had adjuvant treatments with their trial treatment, but considered the influence of these to have been negligible. There were 2 reports of side effects to treatment; both were from the NSAID group.
Schneider et al. (49) selected participants diagnosed with tendinopathy and tested Traumeel S ointment versus 1% Diclofenac Gel. They concluded that, on all variables the homeopathic preparation was non-inferior to the control. On the appraisal of parameters relating to mobility, Traumeel S was found to be better than the control. The limitations they noted were those generally associated with non-randomized observational studies. The authors mention these to be, potential bias of selection, evaluation and “demographic differences between treatment groups” (p.451). However they also mentioned that, by choice of trial duration, the tendinopathy treated was to be considered of an acute level, rather than of a chronic state. The causative origin of the tendinopathy in acute cases is considered to be largely caused by external factors to the patient, rather than to be coming from internal causes, which is in part the case in chronic cases (49). The researchers reported of just one case of side-effects to treatment, this came from a participant in the control group; the Diclofenac group. Compliance and tolerability of both treatments was considered high.
“As effective as conventional medicines” (p.22) stated Schneider et al. (29) in the conclusion to their trial testing Traumeel versus conventional medicines for injuries of an acute nature and trauma. Their results also indicated that Traumeel was a secure preparation of better patient toleration than conventional treatment. Adverse effects were only reported in the control group receiving common conventional therapy for these types of injuries. The authors rated the non-randomised nature of observational trials as one aspect of possible bias. Yet, in terms of selection bias, they considered the influence on the trial outcome to have been improbable due to the lack of relevant differences of the groups at baseline. They further gave some consideration to the healing nature of acute inflictions, as these naturally have a tendency to improve by themselves; therefore the researchers suggested future research to include the rate of recovery from symptoms. They also pointed out that a larger sample size would have delivered more information on the “understanding of the appropriateness” (p.26) of the different medicines used in the study. Participants of this study were allowed to use adjuvant treatments. In the homeopathic group other homeopathic interventions were permitted, while in the conventional group other conventional treatments were allowed. Participants could also have applications such as “functional treatments, compressions etc” (p.23).
Trial quality
The trial quality and reporting of the studies included in this review, according to the respective consort statements for RCT (37) and observational studies (39), ranged from extensive to poor.None of the trials followed and fully met the criteria outlined by the respective statements and as such all boasted gaps in reporting. Likewise, details pertaining to homeopathic specifics were not always described sufficiently as recommended by the REDHOT guidelines (38) for the conduction of homeopathic investigations using the RCT as investigative model. Flaws in conduction reduced trial quality further.
Of the 11 trials here considered, seven produced results that did not provide evidence of effectiveness of the homeopathic intervention investigated. In part we believe this potentially to be due to the poor and biased reporting of the studies. The most attention to detail was given in the observational studies (29, 48, 49) and the RCTs by Jawara et al. (41); Tveiten et al. (44), Vickers et al. (45) and Vickers et al. (46). Thorough reporting for Vickers et al. (46) included the verification of indistinguishability of their placebo and homeopathic remedy, and the preservation of blinding which was reviewed by seven independent researchers!
Raschka and Trostel (26) and Tveiten et al. (43) provided only little detailed information relevant to their trials. Tveiten et al. (43) failed to report eligibility criteria of their participants and included only male participants. The latter criterion made their sample representative of only a male population, giving no account of a female sample. The authors did not explain or show their power calculation for the initial sample size they intended to use and, out of “practical and financial reasons” (p.3631), reduced this sample size from 60 to just 44 participants. At the same time they failed to report of the funding for their trial. Furthermore they produced a measurement gap in the laboratory parameters as blood samples were not taken the day after the race. Tveiten et al. (43) also made the suggestion for future research participants to be screened for their responsiveness to Arnica, prior to their randomisation into groups. This though, would make their sample unrepresentative as only participants would be selected for inclusion that presented with a susceptibility to the homeopathic treatment with Arnica. Raschka and Trostel (26) gave no details of how their participants were recruited and selected their sample from only a small age range with the reported mean age being 28.3 years +/- 4.6 years. This sample is one that can hardly be considered representative of a broad width of the population, and beyond this is also restrictive in number of participants of just 30 by cross-over. They further did not consider the customary exercise regimen the participants possibly were accustomed to, nor did they mention a potential influence of this on outcome. The form of exercise used to induce muscle soreness was one of only short duration which could possibly be viewed critical for such an investigation.
Plezbert and Burke (42) and Schmidt (28) described their studies in moderate detail. In Plezbert and Burke (42) sample size was probably the greatest flaw to their trial. They had only recruited 20 participants and failed to state details of recruitment. Like Schmidt (28) and Raschka and Trostel (26) above, Plezbert and Burke (42) had not given any consideration to habitual exercise of participants and a potential influence of this on outcome.
The greatest details of homeopathic reporting, of relevance, as judged by the REDHOT guidelines (38), was delivered by Jawara et al. (41), Vickers et al. (45) and Plezbert and Burke (42). The observational studies (29, 48, 49) gave lesser descriptive accounts of homeopathically relevant information of the trial substances used, such as potency, method of dilution, or pharmacopeia followed. By the nature of the interventions trialled, these details may be considered of a lesser relevance to these studies, as it is unlikely that a manufactured complex product will be subject to comparison in research to a treatment complex of the same remedy and potency combination. The preparation Traumeel, under investigation in these studies, is a protected brand, exclusively registered with its manufacturer, Heel. In the case of an investigation using a single remedy, on the other hand, such details are essential, as there are many different producers of single remedies that use different production mechanisms and techniques, and follow different pharmacopoeias.
Bias and Flaws noted during review
Sample
Sample size in the included trials was mostly too small (Table 1). Only one trial reported a sample-size calculation prior to conduction of the presented study (46), while Jawara et al. (41) and Vickers et al. (45) produced such calculation for potential future investigations. Only 5 trials were sufficiently powered to allow representative conclusions from the data (28, 29, 46, 48, 49).
Recruitment
Jawara et al. (41) and Vickers et al. (45) recruited their participant sample from hospitals. Jawara et al. (41) in their introduction indicated that the diagnosis of DOMS would permit recruitment from the general public, yet they have recruited their sample from the environs of a hospital only. Albeit that they were including workers, students and nurses, this is not necessarily a sample representative of the general public and as such the trial outcome cannot permit a generalisable conclusion for the broad width of the population. Vickers et al. (45) included “staff and health professionals” (p.305) from the Royal London Homeopathic Hospital, which likewise is a sample unrepresentative of the general public, in particular so, as they were recruiting participants from a homeopathic hospital to conduct a homeopathic trial. This could affect result, as patient expectations, experiences and consequently responsiveness to trial interventions could cause prejudiced subjective interpretations (53, 54, 55).
Short duration exercise
The exercises chosen to induce symptoms in participants may contain further potential to bias outcomes. Potential participants’ preparedness to take part in a trial, as noted by the authors in Vickers et al. (45), may be influenced by the trial exercise regimen. Participants may be discouraged from participation by the type of exercise they are asked to undertake in the trial. As a consequence this may lead to a reduced sample size. Yet, the regimen holds further potential to bias outcomes. The exercises selected, may be viewed critically with respect to the duration and the differing extents of physical activity conducted by the participants. Results could be prejudiced by this variable as the length of exercise conducted could influence the severity of symptoms reported as well as impact on duration of restitution (30). In this context therefore it needs to be noted that the trial by Vickers et al. (46) included participants from 114 races of different lengths ranging from 2 to 50 miles, while participants undertook races of 42km length in Tveiten et al. (43) and Tveiten et al. (44). Participants of the studies by Jawara et al. (41) and Vickers et al. (45) used bench stepping exercises, Raschka & Trostel (26) used concentric weightlifting, while Plezbert and Burke (42) trialled their participants following eccentric exercise, an exertion that Cheung et al. (31) describe to be an action “characterised by an elongation of the muscle during simultaneous contraction (p.147)”. The concentric exercise by Raschka & Trostel (26) described an action of shortening the muscle in the contraction (56). For this review therefore, the latter exercises must all be considered short duration exercises in comparison to the marathon races undertaken by participants in the former trials and studies.
Habitual exercise
Likewise, an uneven distribution of participants’ habitual performance of physical exercise can flaw outcomes. The reviewed trials were conducted by amateurs, mixed groups of amateurs and regular exercisers, as well as more experienced runners or professional sports persons. This may also be viewed critically, and the trial results could be prejudiced by this variable, as here too, the severity of symptoms reported and recovery time could be affected (30). Only Vickers et al (45) and Plezbert & Burke (42) screened their participants for habitual exercise, and only included participants that had no history of customary physical exercise. Tveiten et al. (43) and Tveiten et al. (44) used only marathon runners implying that they were more experience athletes considering the 42km trial distance covered during the Oslo marathon. Tveiten et al. (43) had reported recruiting their participants from sports clubs, such details were not given by Tveiten et al. (44). Jawara et al. (41) noted that future trials would involve exclusion criteria aimed at eliminating this variable by excluding regular and vigorous exercisers.
Preventative prescribing
Apart from the observational studies (29, 48, 49) and the RCTs by Schmidt (28) and Plezbert and Burke (42) all remaining studies that we reviewed, used prophylactic prescribing. Participants were instructed to take the trial substances 12 hours (44, 46) to 24 hours (26, 41, 43, 45) prior to the race or marathon, or the scheduled trial exercise. The adoption of such treatment, by the definition of prophylactic prescribing, is aimed at avoiding possible future symptomatology (57), and as such may have prevented symptoms in these trials from developing. Yet, this approach needs to be viewed critically, as it potentially may have distorted outcomes. In such prescribing it is difficult to confirm the intervention as the influential element leading to a potential absence or alleviation of symptoms. Here numerous factors could impact on the intensity of symptoms a participant possibly may or may not experience, including the afore mentioned habitual exercise of the participants. Moreover, it cannot be ascertained that the remedy prescribed prophylactically, was appropriately selected to match the potentially developing symptomatology probable due to the conduction of the exercise regimen chosen to induce symptoms.
From a homeopathic perspective, this preventative prescribing furthermore remains an issue viewed critically as the principles of homeopathy stress that treatment should be individualized. This can only be ascertained if the patient shows symptoms according to which a remedy may then be selected. Therefore, by prescribing in the absence of such symptoms, patient individuality cannot be respected (5). Prophylactic prescribing therefore can only provide imprecise findings, potentially weakened by the above mentioned and other factors.
Intervention used
It was noticed in Birnesser et al. (48), that the injection solution of the complex preparation Traumeel contained the homeopathic constituents in potencies that were different to oral or topical applications of the same formulation. It was also found that the route of administration of the remedy solution, as opposed to the control substance, differed. While the ˈother-than-placeboˈ control, of conventional NSAID, was administered by intra-muscular injection, Traumeel was given by infiltration. It was not possible for us to make out what the authors meant by infiltrating an injection solution, and how this differed to the intramuscular injection of the control-substance. The definitions we were able to source were inconsistent. One source very simply defined infiltration as the process of bringing a solution into an organic tissue by injection (21). Another source described this procedure as the process of gradually, successively injecting a solution (58). Personal contact to the researchers of this study, for the purpose of clarification on this matter, and in order to determine the purpose of this differentiation in administration, did not yield a response. But as the authors explicitly differentiated this, we assumed that this could inhibit a potential for bias, as it provided another variable to the trial.
Schneider et al. (49) did not restrict the number of applications participants could perform with the ointment to the affected area, and did not direct how participants were to apply the treatment substance. They permitted the participants to select whether they just rubbed the ointment into the skin or covered the affected area in a bandage following topical application. We consider this to be an aspect potentially biasing the results. Whether a topical remedy, is rubbed into the skin, or applied covered by a bandage, can be influential on recovery and treatment perception. In patients where, pressure is experienced as soothing to an injury, a bandage could provide a relieving perception and could result in a positive interpretation of the treatment received and as a consequence could distort a patients’ testimony concerning a treatment effect. In particular where subjective patient experiences are used to interpret the effectiveness of a treatment, this can influence a study outcome. Beyond this, Schneider et al. (49) permitted their participants to have adjuvant treatments during the course of the trial, one of these, was the use of stabilizing elastic bandaging.
What to avoid during the trial period
A factor not to be ignored in a trial on a holistic intervention may be the influence of supplemental treatments or even participant specific customary habits during the progression of the trial. Of the studies here reviewed, such restrictions were formulated only for medical treatments in the form of drugs or therapies (45, 46) and exercises aimed at relieving symptoms, as for example stretching (41, 46). Only Plezbert and Burke (42) indicated to their participants to refrain not only from having any ancillary interventions of medical or manual nature, but even asked them to avoid drinking alcohol during the trial.
When investigating homeopathic interventions, in particular those testing orally administered remedies, there is in this regard an implication to consider. There exists the potential of antidoting, that is, of removing the remedy effect by ingestion of something as trivial as the early morning coffee, for example. According to widespread belief amongst practicing homeopaths, there are many products that are customarily ingested that have the potential to remove the action of a homeopathic remedy (59). While this remains a controversial aspect of homeopathic prescribing, customary habits as for example drinking coffee or brushing teeth with toothpaste containing herbal ingredients imminently before or following the in-take of a homeopathic remedy have been, in some cases, found to remove the remedy effect and consequently in a trial could influence patient perception of the effect of the homeopathic intervention.
Adjuvant treatments
A further complication and possibility of adding flaws to the studies and their outcomes originates from the concession of adjuvant treatments. The observational studies all permitted participants to have supplemental treatments or therapies for the condition investigated by their trial. As such Birnesser et al. (48) permitted their control group to have “oral analgesics or physiotherapy”, and their remedy group to have “further Traumeel injections” (p.121), and oral Traumeel treatment. This gives rise to the question what impact such supplemental treatments could have on the reported effect of the respective interventions, and is of particular interest for the Traumeel group as the potency of the contained remedies in the Traumeel formula differs of injection solution to oral form (Table 3). Birnesser et al. (48) reported that less than 1/3 of participants in the Traumeel group, used adjuvant treatment of mostly the oral remedy. This is a relatively small number, suggesting that the treatment effect reported was indeed due to the complex remedy in injection solution. Schneider et al. (49) and Schneider et al. (29) also permitted adjuvant treatments. Schneider et al. (49) allowed both groups to have manual therapy such as stretching, as well as stabilizing elastic wraps, cryotherapy and ultrasound treatment. Schneider et al. (29) investigated Traumeel, but up front permitted their treatment group to have a combination of “homeopathic products” (p.23), such as tablets and topical applications. They also permitted “functional treatments, compression, etc” (p.23) as adjuvant therapies. Their control group could have “analgesics/anti-rheumatics, anticoagulants, anti-inflammatory” (p.24) and other drugs.
The potential impact of these ancillary treatments should not be left unconsidered, albeit the even distribution in the respective groups being reported. With 69.7% of patients in the Traumeel group and 73.2% in the control group using such adjuvant therapies in Schneider et al. (49), more than two thirds of participants could have report outcomes that were not solely due to the treatment or control intervention; in the least, patientsˈ experience of the extent of improvement could be falsely described. In Schneider et al. (29) 33% in the Traumeel and 31% in the control group received such additional treatments. It cannot be said conclusively that the supplemental treatments, had not impacted on participants and their experiences of well-being or convalescence. Consequently, such adjuvant treatments must be seen as possibly flawing influences on the outcomes reported by the participants.
Incentives
It is considered as problematic, if participants of a trial are offered incentives for their participation. Vickers et al. (46) conducted a prize-draw as incentive for participants while Vickers et al. (45) provided actual payment for participants and stopped recruitment when this funding had been spent. While the former is not seen quite as problematic, a payment made to promote participation in a trial, as in the latter study, is viewed much more critically. This could influence participants’ extent of engagement with, or possibly impact on expressions and descriptions of experiences made during the trial (60).
Ointment control
Schmidt (28) points out, that her trial ointment containing homeopathic Arnica and her control ointment consisted of petroleum jelly. The latter, as mentioned above, has an inherent calming effect that in the event of its application to a sore area could be experienced as relieving. As such Schmidt pointed out that some positive placebo responses in her trial could have been due to the composition of the placebo.
According to the package insert to Traumeel S ointment, the ointment preparation contains Paraffin and Vaseline, both of which are petroleum jelly (61). Schneider et al. (49) investigated Traumeel S ointment versus 1% Diclofenac Gel. They stated that the Gels used in the control group came from different manufacturers, but mentioned that the most commonly used in their trial were “Voltaren Emulgel”, “Diclofenac ratiopharm gel” and “Diclophlogont”. Voltaren Emulgel (62) and Diclofenac ratiopharm Gel (63) also contain as carrier emulsion, petroleum jelly. Of Diclophlogont, as a gel, the ingredients could not be determined. This leaves room for speculation that some active effect of these preparations and of Traumeel S could actually be attributed to the contained supposedly ˈinactiveˈ ingredient, the carrier substance. This could account for some of the positive effects in the placebo or respective other-than-placebo groups. While this is negligible in a trial where both trial substance and control contain the same basic ingredients, in a trial versus placebo control like Schmidt (28) this could pose a problem and could be the cause of some distortion of results. Schmidt (28), did note in her trial that the differences in effects of the trial ointment to placebo could be smaller, as placebo effects could falsely have been interpreted as too great, due to this inherent effect of the contained basic substances.
Funding
The Dove Healing Trust supplied the funding for Vickers et al. (45), theBlackie Foundation for the study by Vickers et al. (46), and the Norwegian Research Council funded the trial by Tveiten et al. (43). Birnesser et al. (48) and Schneider et al. (29) received funding from Heel the producer and marketer of the complex homeopathic preparation Traumeel.
The funding sponsor of a trial may be viewed as a source of bias. Where funding is expended from resources related to the intervention under investigation, critics may imminently see an intention, by the financial supporter, to extract an advantage from the research. The Norwegian Research Council can be considered an independent source of funding with no direct connection to homeopathy as it is an advisory body to the Norwegian government (64). The Dove Healing Trust and the Blackie Foundation are respective sponsors of CAM treatments (65), and homeopathic research (66). They are bodies that too may be considered independent from bias, as their monetary funding comes from donations and charity, that is, from independent sources that have no direct relation to any research. What financial givers of these charitable funds possibly are aware of, is that their donations will be expended for homeopathic, or respectively CAM interests. The funding of trials by Heel though may be considered as controversial. In this case, an exertion of influence on findings reported by the trial could potentially be assumed, as the spending on the trial, by Heel, serves to inform of the effectiveness of a Heel-product.
Trial methodology – mode of prescribing
A consideration of primary importance when investigating homeopathic preparations is that of the methodology used to test the intervention. This may have a causal impact on study outcome that at the very root of the research may flaw the trial. The random controlled trial is viewed as the gold standard of scientific testing, but is criticised for not sufficiently acknowledging aspects pertaining to homeopathic practice when used to trial homeopathic interventions. Plezbert and Burke (42), Schmidt (28), Tveiten et al. (43) and Tveiten et al. (44) noted this in their trials. Of the trials included in this review eight are RCTs.
Interpretation and Synthesis
It became evident that review synthesis could only be established with trials that had sufficient similarities. Unfortunately the here included trials boasted more discrepancies than communalities. We therefore decided to exclude from synthesis trials that were of poorer quality, and did not fulfill minimal criteria mutual to the better quality studies. As can be seen from table 4, the selected studies had to report significant positive outcomes, an adequate sample size, a sample representative of the broader public, a longer exercise duration, had to commence treatment of participants following exercise or diagnosis, and as such had to be least possibly flawed. These criteria delivered only 4 studies that qualified for synthesis.

None of the trials used an individualized mode of prescribing. All have implemented a generalized approach. This is more commonly the practice in conventional treatment where the diagnosis indicates the prescription and not the idiosyncratic symptomatology expressed by the individual patients. In the case of the observational studies (29, 48, 49) such generalized prescribing is more justified an approach than for prescriptions of single homeopathic remedies. The presented observational trials all tested a homeopathic complex preparation, Traumeel, and as such do not fall under the criterion of individualised homeopathic prescribing. Complex formulations are designed to be prescribed in a generalized manner as the number of ingredient remedies are aimed at meeting a broad spectrum of symptomatology expressed for a specific condition. They can thus can be aimed at a more diverse population (25). Consequentially the selection of a homeopathic remedy, under consideration of the idiosyncratic aspects of each individual patient, is much simplified (25), as there is less requirement to be specific. This is a procedure violating the homeopathic principle of individualization as postulated by Hahnemann (5), and is considered an entirely different approach to homeopathy that had initially been put into practice by Pastor Emanuel Felke (67, 68). As such, this method is today being vastly practiced and recognized as a stand-alone therapeutic range of homeopathic treatment.
Of the reviewed trials only one RCT on a topical application of Arnica in two different potencies (28), and the three observational studies (29, 48, 49) investigating the homeopathic complex preparation Traumeel versus conventional treatments, were able to produce results favourable of a homeopathic treatment for the reported indications. Schmidt (28), Schneider et al. (49) and Schneider et al. (29) investigated topical applications of the trialed homeopathic preparation, while Birnesser et al. (48) investigated the effectiveness of an injection solution.
Jawara et al. (41) stated that their trial showed “moderate support” for the homeopathic treatment, but correctly acknowledged that their trial was insufficiently powered for results to demonstrate a statistical significance of their outcomes. Other than Birnesser et al. (48), Schmidt (28), Schneider et al. (49) and Schneider et al. (29), the remaining trials all administered their treatment preparations via the oral transmucosal route of administration. Their outcomes of effectiveness were all reported as not statistically significant. The trialled remedies were of different potencies, and all were taken orally by the participating subjects of the studies. This raises doubts on the effectiveness of orally administered homeopathic remedies.
Even though adjuvant oral or other treatments were permitted as complement to the Traumeel investigations, as stated above, it appears from the results, that the route of administration of a homeopathic remedy may be a yet insufficiently considered variable in research into the effectiveness of homeopathic treatments. In a survey previously conducted by the reviewers of this paper we had interrogated practicing homeopaths about their beliefs and experiences concerning the routes of administration and the prevalent use of the oral transmucosal intake of homeopathic remedies. The outcome implied that it was not believed that the route of administration impacted on the effectiveness of the homeopathic remedy. But this interrogation was underpowered and we concluded that further research would be advisable (69).
The here reviewed trials made us notice that the potencies in which remedies were prescribed, could yet be another variable influencing effectiveness in relation to the route of administration. As illustrated in Table 2, a variety of potencies had been investigated by the reviewed studies. Arnica in X/D1 and C6 as reported by Schmidt (28), was found to be effective in topical application. Arnica is one ingredient of the complex formulation Traumeel, as noted in Table 3, and is contained in the topical ointment in a potency X/D3 as opposed to X/D2 in oral and injection solution. Could the potency influence the uptake of the remedy and as such flaw outcomes of investigations due to inappropriate potency for a particular route of administration?
The topical applications in these trials used potencies that still contained material substance of Arnica. Homeopathic preparations only loose the content of material ingredient by surpassing Avogadro’s number, at roughly C12 (4). Therefore, the trial by Raschka and Trostel (26) might support this theory. They reported that their investigation showed no effect of oral Arnica in a similar material potency of X/D4. Yet the topical applications of Schmidt (28) and Schneider et al. (29, 49) showed effectiveness. This interpretation though should be viewed with caution as the trial by Raschka and Trostel (26) has been poorly reported and was flawed particularly by the small sample size, as noted above.
In the light of the outcomes reported in this review, the argumentation here considered, could be one of relevance for future investigations into the efficacy of homeopathic remedies. Future research could focus on appraisal identifying the most appropriate route of administration of a remedy, possibly giving rise to new aspects to consider, or to reject the argument that we here propose.
It is also striking that Schmidt (28), the observational studies by Birnesser et al. (48), Schneider et al. (49), Schneider et al. (29), and Plezbert and Burke (42) did not resort to prophylactic prescribing. Plezbert and Burke (42) investigated Arnica C200 given orally following eccentric exercise. Plezbert and Burke (42) concluded that Arnica in a high potency is not effective for the investigated indications.
All other trials had their participants begin taking the trialing substances prior to conducting the exercises. Considering that all other studies included in this review concluded that the trialed homeopathic remedies and formulations were ineffective for the respective indications, could suggest that prophylactic administration may not be a means for effective homeopathic prescribing.
Some consideration though, has to be given here to the prescribing by Schmidt (28) who did not indicate for participants to apply the trial substances prior to the physical exertion, but who suggested application immediately following a race. She also included participants that only had to expect to have symptoms from overexertion, and instructed them to apply the ointment immediately at the end of the race, and as such before the onset of the ˈanticipatedˈ symptoms. This is also a form of prophylactic use; in the least for some of her participants. It is therefore noteworthy that in her trial, a mix of non-prophylactic and prophylactic administration may have taken place.
Beyond this it is also notable in the context of this synthesis, that Schmidt (28) and Birnesser et al. (48), Schneider et al. (49) and Schneider et al. (29) had the largest sample sizes for their trials (Table 1). Only Vickers et al. (46) exceeded their number of participants. Vickers et al. (46) had tested oral Arnica X/D30 on marathon runners participating in races of different lengths. They concluded their intervention to have been ineffective.
It is reported throughout the trials and studies reviewed, that homeopathic remedies whether single or complex preparations, were very well tolerated and patients were extensively compliant to these prescriptions. As the observational studies show, this tolerability and compliance was even beyond that commonly acknowledged for conventional treatments. Likewise the reports of adverse effects in the RCTs, by the even distribution between treatment and placebo group, suggest homeopathic remedies provide little to no discomfort and participants find their handling acceptable.
Consequently, should finally a model of testing holistic interventions be identified that takes into account the non-specific aspects fundamental to holistic treatment approaches, that is at the same time sensitive to the route of administration and the potency prescribed, then future reports of evidence of effectiveness of homeopathic treatments could dismiss the prevailing criticism.
Our results showed no agreement on a definitive positive or negative outcome reporting effectiveness of homeopathic interventions. This as stated further above is rendered difficult by the diversity of indications, remedy potencies, research models used and outcomes reported. None-the-less the few similarities and the vast differences noted in the different trials permit following critical argumentations:
The homeopathic complex preparation Traumeel, of ingredients in mother-tincture and decimal potency, and the single remedy Arnica in X/D and low C potency are effective for sports related injuries if applied topically following physical exertion or exercise. These preparations furthermore have a profile that describes them as very well tolerated and patients as very compliant to their use.
All trials were undertaken using a generalized mode of prescribing opposing the fundamental homeopathic principle of individualization. Excluding the investigations into complex preparations from the following argument, as their nature of prescribing permits a more generalized approach, all other trials could be flawed by this very approach of prescribing and consequently at the very core could be falsified in their outcomes.
Furthermore, justified critique of the research models used to investigate homeopathic interventions, in particular of the RCT, and of the placebo used as control make a consensus statement on the effectiveness of homeopathic interventions difficult.
DISCUSSION
Our review has shown that something just isn’t right with the research into holistic therapeutic approaches and into homeopathy in particular. Further to the flaws and weaknesses identified in the individual studies above and as a consequence to them, more bias may occur.
We have noted above that funding may be an issue for homeopathic research. This may occur where reported outcomes arouse the suspicion of a potential exertion of influence from a financial sponsor, seeking to pursue his own monetary or other interest (70). This is no issue pertaining solely to the alternative practices or homeopathy in particular. It is in fact one that is common to all sectors that are conducting research (70).
For homeopathy, there exists the problem that there is little priority from outside the homeopathic field, to see research undertaken investigating homeopathic interventions. Consequently there are few independent organizations that are willing and interested in, supplying funding for investigations into homeopathic preparations (71). Therefore, financial supporters of homeopathic research frequently come from domains that somehow stand in relation to homeopathy, such as the sponsors who funded the trials included in this review, the Dove Healing Trust, the Blackie Foundation, or even the producers of remedies such as Heel.
Yet, further complications associated with the expenditure for research into homeopathic investigations may arise. Trials are expensive to conduct and larger sample sizes imply higher expenses and costs. Two of our included trials reduced their participant number because their funding had been expended (43, 45).This reflects the monetary problem largely prevalent to all research conducted in complementary and alternative medicine as we have mentioned.
There are from this, implications on other forms of bias that could possibly occur in a trial or study. For example, where the participant numbers of a trial are greater, publication bias is less probable. (72, 73). The likelihood that a large study reaches publication despite negative reporting is greater, where expenses and trial associated efforts have been extensive due to the larger sample (70).
But there is much more potential for flaws and weaknesses of trials investigating homeopathic interventions, and these arise at the very foundation of the investigation. Where the fundamental principles of a treatment approach are ignored or are inappropriately applied, reported outcomes cannot reflect the true effects of the intervention tested. Furthermore, where the methodology used to investigate a treatment, itself, boasts discrepancies and is for example, ignorant of most basic aspects fundamental to human perception, and of methodological features that have implications on conduction and therefore on outcomes, such as measurement parameters and trial control, a trial is doomed to falsification even before it is undertaken. This is the case with the application of RCTs for research on homeopathic interventions.
A closer look at methodology and investigations into CAM
In the hierarchical structure of research designs, a randomized controlled trial stands higher than an observational study. The RCT is seen as the tool of highest quality for investigations of efficacy of interventions, and as such holds the top position of the hierarchy, whereas the observational methodology is considered of a lesser standard, and as such lies lower in the hierarchy, below this ˈgold standardˈ, the RCT (74).
Observational studies are believed to be of less rigidity and as a consequence are thought to produce more chance for errors than methodologies of a higher rank in hierarchy (74).
There are however, important aspects that suggest that observational studies may have considerable advantages over randomized controlled trials (75). Observational studies are believed to reflect a much more realistic clinical situation, providing a greater generalisability than does the RCT. Observational studies can run for a much longer follow up phase, may include a much larger participant number, and costs are not as high as those for undertaking a randomized controlled trial (75). A major aspect of observational studies, viewed as a disadvantage, is that of potentially producing outcomes showing treatment responses that have been exaggerated in their strength (75).
The investigations by Benson and Hartz (75), and Concato et al. (74), looked at the qualitative implications of observational studies compared to randomized controlled trials, and did not produce results confirming the assumption that observational studies are qualitatively less rigid than RCTˈs. From their findings the authors could also not substantiate that outcomes of the observational research model are necessarily inflated. In fact, Concato et al. (74) concluded, that if the rigidity in the conduction, and that of participant criteria are sufficiently similar, observational and randomized controlled trials may provide similar outcomes.
The observational studies found for this mixed systematic review, were of a much higher quality of reporting and more rigidly conducted than were most of the RCTˈs that were included. In fact, we included only the three observational studies and just one randomized controlled trial in our review synthesis, as we considered it inappropriate to draw a synthesizing conclusion taking into account investigations that had been badly conducted and were insufficiently detailed.
As the observational research method gains credibility compared to the RCT, following the investigations of Benson and Hartz (75), and Concato et al. (74), it becomes clear that the common notion that the RCT abolishes all bias and delivers only rigid outcomes, is false (76). RCTˈs have been known to produce results that in practice were eventually found to have been lacking substantial considerations, so for example, indications of unfavorable and favorable impacts that were found to be unrelated to the actual treatment effect of the investigated trial substances (77). The gold standard of trial methodology, as such exhibits flaws, because its design simply is not structured to identify certain features that are related to the treatment effect, but are not directly measurable by the chosen outcome parameters.
The RCT is a methodological design devoid of parameters taking into account impacts, such as non-specific effects, that may likewise be as influential on trial outcome, as the anticipated effect of the treatment investigated (55). Therefore, the gold standard of medical research cannot be considered a flawless testing tool that produces only firm and correct statements (74, 77). The RCT too has limits, and investigations into interventions of CAM, clearly illustrate such limitations (76, 77).
RCTˈs for investigations into homeopathy
How appropriate can it be, to employ a research tool that at its very foundation is inadequate for investigating treatment approaches that encompass holistic views that are sensitive to nonspecific factors, and do not follow clinical disease diagnostics, but underlie alternative therapeutic principles?
The conventional medical practice relies mainly on the specific factors of a treatment, namely the management of drug intake and the medicinal effect (55). This orthodox approach engages only superficially with the patient, leaving little room for individual patient concerns or a patient-centred therapeutic relationship. On the other hand, the holistic treatment approach of many complementary and alternative medical therapies, exhibits unspecific factors that have been found to be influential on the improvement patients make following such treatment (55). The individualized nature of such therapies and the associated deeper engagement with the patient, before, during and after the treatment arouses these factors, making them potentially therapeutic to the patient albeit being largely unrelated to the actual treatment intervention (55).
The randomized controlled trial is a research tool aimed at identifying those treatment impacts that are characteristic of an intervention (77). As such, this methodology answers the questions that are fundamental to orthodox medicine, as mentioned above, as it focuses on identifying the medicinal impact of an intervention, the so called specific effect of a treatment. This method has advanced to become the gold standard of research in orthodox medicine (77), and evidence based medicine has postulated the RCT as the best tool for providing reliable evidence of trialled interventions (76, 78). As a consequence the therapies of CAM too feel forced to conduct research using this method to attain credibility for their therapeutic approaches (77). Yet where the RCT is used to investigate interventions of CAM, its trial design is inappropriate and “negative results are almost inevitable” (77 p.73).
A fundamental principle underlying the research method of the RCT, is assignment by chance. Participants are randomly spread to a treatment and a control group in the aim of assuring that variable factors, that potentially could impact treatment outcomes, besides the intervention to be tested, are balanced between groups (76). Yet this comprises one aspect that potentially impacts the outcome of research into homeopathic interventions.
Randomization – a complication
The tenet of randomization is the blinding of participants to their group allocation and consequently to the treatment, be it active agent or placebo (79). While it is aimed at reducing bias, such as that of selection, it poses complications that cannot be ignored in particular when investigating CAM treatments (79).
A trial situation is a simulation, and as such rarely reflects a normal treatment practice (36). In a trial, participants are aware of the possibility that they will not be in the treatment group, but may actually be receiving a placebo instead of the active substance. This distorts the true clinical situation patients in practice are subject to, and may therefore lead to biased outcomes, thereby reducing the external validity of the trial (36, 79).
Such studies, including a placebo control eliminate factors such as patient choice, and hence may produce opposition of the participant toward the trial treatment they may possibly be receiving (80). This may instigate a situation that Corrigan & Salzer (79) term “diminished motivation” (p.110), which of course has impact on the internal validity of a conducted study.
At the same time such a study may become biased at the very start, even prior to the randomization stage, when potential participants, because of a categorical preference for the trial intervention, may decide to not participate at all for fear of being allocated to the control group (79). This may reduce sample-size and in consequence may negatively impact external validity. This could have been a possible influence on the reduced number of participants that had been prepared to take part in the trial by Vickers et al. (45) that we included in this review. Vickers et al. (45) had mentioned difficulty recruiting participants.
Corrigan and Salzer (79) point out that presently there is no replacement for randomization as it still best restricts confounding impacts, yet they also mention that it needs to be viewed critically, and implications should not be ignored when considering designs for future research.
Specific versus non-specific effects
As we have pointed out above, non-specific effects do influence patients. They may impact on their comportment in a specific clinical situation, and they may have an influence on how they experience and perceive the effect of a treatment intervention (80). Such non-specific factors are considered to come from concerns, beliefs, expectations (53, 55) or subjective experiences of the patient (54). These effects are especially prevalent, recognized and acknowledged as influential aspects, in the CAM practices (55). The conventional medical approach is not particularly sensitive to such factors. From a conventional medical perspective, the specific parameters, the medical impact and handling of a drug substance, are considered the only treatment influence, and the unspecific factors are considered to be attributes of a placebo effect (82). In consequence to this, the research methodologies of conventional medical practice are indifferent to such effects. They have not been designed to investigate such non-specific parameters.
Concato et al. (74) state that “the experimental protocol for therapy may not be representative of clinical practice” (p.1891); but for investigations seeking to answer questions on homeopathic effectiveness, this very difference must be overcome. The multiple non-specific factors influencing a homeopathic consultation need to be taken into consideration in order for the treatment impact to become measurable. Walach (82) states, that the non-specific effects are the foundation on which the specific factors are perched. Therefore the non-specific effects cannot be subtracted from the treatment influence made on an individual receiving an intervention. The RCT though, assumes exactly this, believing that the non-specific factors are equal in trial groups, treatment and placebo, thus leaving only the true effect of the intervention as a measurable outcome (83).
As a result of this discrepancy, where an investigation into homeopathy uses the RCT, only potential specific effects of the intervention are sought to be discovered. As these though cannot be found for homeopathic interventions using this research design, for reasons that we will elaborate on in the context of this discussion further below, homeopathic treatments have been rated as ineffective and criticism of, and the opposition to this treatment approach have been nurtured.
The problem with Placebo – It’s paradoxical
The ignorance over the role of non-specific effects in the treatment and recovery of patients in conventional research, boasts an inherent complication that is too often left unconsidered. The RCT, the random controlled trial, requires a control treatment against which the active medicine is tested. This control most frequently is a placebo, a sham intervention which is a substance lacking the active ingredient, yet otherwise identical to the trial material investigated (78). Especially when using the RCT to investigate homeopathy or other alternative interventions, the use of placebo as a control must be viewed critically. The placebo control may actually exhibit a principal discrepancy for the application of the randomized controlled trial to investigations into homeopathic treatment effects. Walach (80) states, the placebo is a flaw, if used to trial versus CAM interventions, and Horn et al. (78) state that “…with very few exceptions, sham-controlled trials have numerous limitations, are poorly executed and improperly interpreted” (p.1).
The main reason for trialing against sham or placebo is the belief that this will diminish bias (78). Unfortunately, it appears that although potential bias of the participant may be reduced, by this assumption, other bias may actually be introduced into the trial (78).
Placebo has always been thought to be an inert procedure, lacking any active excitatory or stimulatory potential, and as such has been viewed as inept of instigating any psychological impact on a research participant (78). As such therefore, any psychological impact has been assumed to be associated with only the trial treatment given. But as Horn et al. (78) and Enck & Klosterhalfen (83) point out, this has never been ascertained by research.
In a trial situation the participant finds himself under conditions that are sought to mirror actual clinical circumstances, yet rarely do. As we have pointed out, any participant of a trial has to be informed in advance that potentially he or she will be receiving a sham intervention rather than the drug that is to be tested. This is a very different state of psychological awareness for a patient and is much different to any clinical situation that a patient customarily experiences (81), and as we have mentioned above this may influence a participantsˈ perception of an effect and may as such flaw the experiences that he or she reports of a treatment (81). Consequently the trial results could be falsified.
The paradox
It is a widespread belief that a placebo is a substance that has no specific impact (78) and that unspecific factors are of the same dimension in either trial group and thus can be neglected (82). It is furthermore an adopted creed that solely specific effects are of therapeutic value (76). Yet, non-specific factors, as mentioned above, have the potential to influence participants. Where the impact of the control may fall under influence of participant hope, outlook, lifestyle, comportment (82), or “behaviours embedded in medical rituals” (84 p.1), possibly also exhibited by the therapist (82), the control cannot be considered to be inactive. Consequentially trial findings cannot report unbiased outcomes. Therefore the use of the placebo control in trial situations is paradoxical.
To utilize a control presupposes that the findings of the placebo group are no different than the results of a non-treatment arm. Only if there is no effect from the placebo, that is, the control is inert, only then can the findings in the treatment arm of a study be fully acknowledged to the tested intervention (78). Unless therefore, there is certainty that the placebo control is neutral, devoid of specific and non-specific effects, the randomized controlled trial exhibits an elemental weakness. This variable can only be accounted for if a study were to include a third trial arm that consisted of a control group that was left un-treated (78), allowing the identification of potential non-specific effects by comparison of outcomes to the placebo group. In Schmidt (28) above, for example, such a third arm could have clarified on the impact of the placebo on measurements. The researcher had chosen to use petroleum jelly as control substance. She considered this control substance to have an inherent treatment effect that could falsify findings in the placebo group, and consequently could have implications on the overall outcomes of the trial. Comparison to the third trial arm could have delivered a numeric measure of this placebo impact, which could then have been subtracted from the treatment findings to produce data caused solely by the intervention. Yet, for this trial by Schmidt (28) though, a non-treatment arm became unnecessary as the trial substance too contained the same type of jelly as a carrier substance. Contrary to this description of the paradox, in Schmidt (28), the impact solely from the basic jelly therefore, could be expected to be the same in both groups. Only if Schmidt (28) had wanted to measure this inherent effect, would the third arm have been a tool to provide such findings, but this had not been an exploration of her trial.
Recommendations for research methods
Efficacy over effectiveness – another controversy
In the sphere of scientific research it is assumed that without proof of efficacy there is no effectiveness of an intervention (85). The specific effect is therefore required before a treatment is judged as effective (86). Yet many of the practices of CAM, in clinical scenarios, are appreciated for their effectiveness, albeit the lack of the provision of evidence of efficacy. Walach (86) in this relation points out, that for the practices of CAM “their way of achieving efficacy is non-specific” and that “part of their effectiveness is covered by most of their nonspecific effects” (p.214). Where the non-specific effects therefore, are as high to render a treatment effective, a trial methodology seeking to reduce these factors for only specific impacts, goes past the true evaluation and recognition of a potentially successful treatment approach (83, 86).
Placebo versus Other-than-Placebo
Therefore, a CAM treatment that consists to such extent, of non-specific effects, is unsuited for testing with the RCT. The randomized controlled trial is a model seeking to reduce placebo effects while emphasising on the identification of specific impacts only. Walach (80) therefore suggests a different approach to researching CAM practices and postulates trialling the alternative intervention against a conventional treatment. Although an intervention may show efficacy by exhibiting specific effects that are significantly increased compared to placebo, the conventional treatment may still be less effective than a CAM therapy with high non-specific measures, albeit lacking superiority over placebo (80, 87). Walach (80) calls for studies that are “varied and multifaceted” (p.1141) and stresses that these should reflect “real-life” (p.1141) experiences, something that is obliterated by the use of randomization, as we have noted above.
New models for research – recommendations taken from the literature
The practices of CAM, including homeopathy, have to be subjected to research in order to provide evidence that they are without harm and have an actual impact (77). Such trials and studies must concentrate not solely on specific effects, but need to investigate all effects pertaining to the intervention, also those that are non-specific (77). This cannot be established by just conducting a single trial using one research model, but must be acknowledged and evaluated on “all levels of evidence” (77 p.74). Walach (82) points out, that in order to recognize the collective effectiveness of all impacts of an intervention, the findings of numerous trials employing different research methodologies have to be appraised and evaluated. He stresses, that only following such synthesis can the real effectiveness of an intervention be identified.
Every methodology used in scientific research has advantages and shortcomings. To answer a certain question, the trial method has to be selected such that the potential findings are best suited to respond to the query. Yet seldom are the system inherent weaknesses of a trial without effect. Another study, using a different methodology may therefore be needed to investigate for these impacts. As such, in synthesis with the former trial, more accuracy can then be attributed to the outcomes (76). Walach (76) therefore suggests a different non-hierarchical structure for the evaluation of research. He advocates a circular model where a trial methodology investigating efficacy (specific effects) is paired with a method that aims to inform of factors describing effectiveness (non-specific effects). With this model, he postulates, that it can be achieved to evaluate a trial situation in a clinical context that is sensitive of individual patient choice and to the impact of the therapeutic relationship, while at the same time it provides thorough scientific proof (76).
The impact of homeopathic principles
Routes of administration and potential difficulties involved
We could not identify in the literature or current research, material that investigated the routes of administration of homeopathic remedies and the potential influence potency selection could have on this. As we mentioned above though, we had conducted a survey some time ago with the aim of investigating the beliefs of homeopaths concerning the effectiveness of the different ways of remedy intake (69). From the results it became evident that practicing homeopaths did not believe that the remedy action was influenced by the route of administration of the remedy.
A second survey, for the same paper, among European producers of homeopathic remedies showed that in homeopathic practice the oral route has large predominance over other routes. While this is also true in conventional medical practice (88), the investigation of this topic in allopathic literature exhibited the limitations of this pathway of remedy ingestion by way of the digestive processes that occur in the body (88 – 90). Decomposition processes are known to reduce the amount of active ingredient that eventually can unfold its curative action once the drug has reached the intestinal tract where it is then absorbed (91).
In the case of homeopathic remedies that are usually recommended to be retained in the oral cavity until dissolved, for absorption via the oral mucosa (92, 93, 94, 95, 96), their action could potentially be disrupted by foods, drink or other substances taken into the mouth (88, 97). As such, therefore there could be concern, that the effect of a homeopathic remedy could be ˈantidotedˈ accidentally by having a drink or brushing teeth following the administration of a remedy.
Hahnemann himself only mentioned such consideration in his writings, for substances with medicinal properties (5, Aph. 259 & 260). Yet recommendations, as for example, to avoid drinking coffee right after or before taking a remedy are widespread amongst practicing homeopaths and are frequently down to experiential reports (59). Such precautionary measures therefore, may be relevant for participants of trials, as no definitive answer has yet been found to the question of whether or not such effects exist.
The above trials had not considered such impacts. Yet, following a strenuous physical exertion as a marathon for example, athletes may seek to nurture their devitalized physique and are likely to ingest food and drink aimed at nourishing their exhausted constitution. As a consequence to doing this, the athletes could influence the action of an orally administered homeopathic remedy. Therefore, this could have an impact on such prescriptions given orally right after a race or following exercise.
Likewise this could also play a role in prophylactic prescribing, ahead of exercise and exertion where customary eating and drinking habits of participants could reduce the remedy effect. The trials of this review, as mentioned above, practiced prophylactic prescribing for all oral interventions. Only the topical applications, and that of injection, were conducted following a race or after clinical diagnosis had been established.
The route of administration could also play a role in the case of adjuvant treatments. These had been permitted in the trials by Birnesser et al. (48), Schneider et al. (49) and Schneider et al. (29). If, what we presume in relation to the route of administration holds true, then the adjuvant treatments could indeed play a role in the reported impact of the trial interventions. For example, in Birnesser et al. (48), adjuvant oral treatments with Traumeel were permitted in the treatment group as an addition to the trial intervention of Traumeel as an injection solution. If therefore, the oral route were found to be more effective than an injection, for example, the extent of effectiveness reported to be due to the trial intervention could be falsely interpreted, because in actual fact the impact could be due to the adjuvant treatment. The same holds true for the trial by Schneider et al. (29). Here the researchers not only permitted adjuvant treatment, but investigated Traumeel as a mono-therapy or in combination with other homeopathic prescriptions. This combination treatment consisted of oral and topical remedy administration. Therefore, it can be pointed out, that this issue is one that should be investigated, as it could exhibit potentially biasing influence on a trial investigation.
If this impact were found to be prevalent, it would next be essential to identify what treatment effect different potencies have, depending on the route by which the remedies are administered. This has never been established, nor has been subject of an investigation. The underlying concept of why the ingredients of the complex preparation Traumeel have different potencies, in oral and topical form or as an injection solution, our personal contact with Heel, the manufacturer of Traumeel, could not satisfactorily clarify (Personal communication 2013 February 25). This leaves space for speculation as it adds a potential variable to investigations into homeopathic interventions. We therefore believe it is justified to pay attention, to homeopathic interventions, their potencies, their route of administration and the potential influential factors on remedy uptake in future trials and studies. The findings of our review have raised these issues, and we find them to be potential variables in need for further attention when investigating homeopathic interventions. We recommend separate research in order to clarify on the extent of impact of these aspects of homeopathic prescribing.
Preventative prescribing and the question of generalized or individualized
As we have mentioned above, all but 4 trials of this review used prophylactic prescribing. The majority of studies asked participants to take the trial medication, active remedy or control substance, in the absence of disease symptoms, prior to the potentially symptom inducing event. The aim central to such administration is the avoidance of a future infliction with an ill-making agent, an infection or a disease (57). This though is a controversial treatment procedure amongst practicing homeopaths as one of the principal dicta fundamental to the practice of classical homeopathy opposes such preventative prescribing. In Aphorism 82 of the Organon Hahnemann (5) stated that:
It remains, for literacy of the indication of each disease (psoric) to be healed, for the homeopathic doctor an indispensable duty to accurately record the plumbable symptoms and idiosyncrasies…
…there cannot be a true healing of this or all other diseases, without strict individualisation of each case of illness.
The emphasis on patient individuality and idiosyncratic case-taking is therefore manifest for many practitioners within Hahnemanns own writings, and according to this Aphorism is irrevocable for classical homeopathic prescribing. According to this statement of Hahnemann in the Organon, prophylactic prescribing must most certainly be deemed in disaccord with his postulations. Yet to advocates of a preventative approach of prescribing, the justification for a generalized treatment mode, as it is common in conventional medical practice, is delivered by Hahnemann (5) in Aphorisms 101 and 102 of the Organon. There he outlines the procedures for the homeopathic treatment of epidemics for which he prescribes the genus epidemicus, the remedy that best matches the characteristic symptomatology of the epidemic (98):
…Each of such Collective-diseases exhibits only upon closer observation of multiple cases the epitome of its symptoms and signs…the diligently exploring practitioner can even find the true state and the characteristic picture from the first or second patient…and can then already find a matching homeopathically adequate curative… (Aphorism 101)
…the outlined disease picture becomes increasingly complete…it becomes more characteristic and encompasses the peculiarities of this Collective-disease…the subsequent cases will confirm the accuracy of the selected remedy or will point [him] to a even better matching homeopathic remedy, the simillimum… (Aphorism 102)
…All who contracted this pestilence have the same disease that came from one and the same source, the entire extent of such an epidemic disease and the totality of its symptoms belong to its cognition… it cannot be perceived just from one sick individual, only from the suffering of multiple ill, of differing physical appearance, can it [symptom complex] fully be abstracted and extracted… (Aphorism 102)
Historically, and from the extensive experiential background it is known that Arnica, for example, is a remedy with a reaction radius that predisposes it for the treatment of injuries potentially contracted from sports or other physical exertion (19). As such, the researchers of our reviewed studies used Arnica because the conditions they investigated belonged to the symptom complex of sports injuries. They treated the trial indications according to the principles laid out by Hahnemann for the treatment of epidemics, and therefore used a generalized mode of prescribing.
Evidence suggests that such generalized and preventative prescribing, in the case of epidemics, is an effective approach. A recent and most famous report of such homeopathic treatment in an epidemic comes from Cuba, where a study performed in 2007 during the annual epidemic of Leptospirosis, resulted in dramatically reducing infections in the high risk population of almost 2.5 million people (99).
Treatment for Epidemics – a homeopathic dictum falsely applied
In the light of such successful application of the Hahnemannian approach we need to raise, for our review, the question if not inflictions with sports injuries fall short of indications that fulfil the criteria of an epidemic. Furthermore we need to ask if not this may be a cause of the negative outcomes. An epidemic, by definition is an infection or contagious disease that spreads rapidly and affects many people (21, 100, 101). Delayed onset muscle soreness, for example, the principal indication investigated by the trials included in our review, can with this definition not be considered a disease of an epidemic nature. The symptoms of DOMS have no infectious or contagious characteristics and are inflictions that by natural course would go away after some time if the inducing exercises were desisted (102). As such therefore, the preventative prescriptions undertaken in the here reviewed trials, are not following the principle of a genus epidemicus. The mode of prescribing used here must therefore be led by a different philosophy, possibly that of ˈgeneralized homeoprophylaxisˈ (98). Yet this is even more controversial, as it, in principal, uses a conventional medical approach to treatment that is undertaken with homeopathic remedies. This concept emphasises on delivering the same treatment to many persons suffering of different ailments (98), and as such poses more discrepancies than the treatment with the epidemic genus. This treatment approach can be related to the orthodox policies of vaccination, and is as such in its own right viewed critically. If this concept though, were used as justification for the preventative treatment used in the trials we reviewed here, the application of this mode of prescribing would likewise be governed by false pretences, and would be equally as erroneous and inappropriate as is the use of a genus epidemicus.
Therefore, if randomized controlled trials do not fit fundamental criteria of classical homeopathic prescribing, their outcomes cannot deliver unbiased conclusions. At the same time, if studies did meet the criteria, and the outcomes would still be lacking heterogeneity, then they would have to be fundamentally flawed elsewhere. The inconsistent outcomes of the studies included in this review therefore neither provide stringent evidence for, nor against effectiveness, but their presentations do exhibit shortcomings and bias, as we were able to report in our findings.
Implications of our findings – a conclusive statement
Our review has clarified that we need to undertake trials and studies with more attention to conduction and reporting. This need has been clearly substantiated. Our appraisal further disclosed discrepancies to investigations trialling homeopathic interventions using the gold standard of scientific research, the RCT. It became evident that the randomized controlled trial fails to address aspects that are principal to homeopathic treatment, and consequently the outcomes of the individual trials cannot provide consensual findings. We therefore require a new methodology for testing CAM procedures, or an investigatory model that permits the drawing of synthesising conclusions, by combining the outcomes from more than just one trial. The latter findings should in turn have been acquired by research conducted using different existent methodologies.
We recommend explorations into the efficacy and effectiveness of the different routes of administration of homeopathic remedies, as our review has pointed to a potential weakness in the evaluation of homeopathic treatments. While experience in practice appears not to know of such influence (69), the evaluation of our findings has evoked significant concern over a possible impact of this on the effectiveness of a homeopathic prescription.
Our findings have also pointed out that, besides the delivery of evidence of efficacy and of effectiveness of homeopathic treatment, trials should be performed that investigate into the principles of the individualized versus the generalized mode of prescribing in order to be able to identify if and how homeopathic prescribing is most efficaciously conducted. Potential information retrieved from such comparative inquiry may likely clarify on some of the discrepancies pertaining to the fundamental homeopathic principles. Up until today such trials and studies are lacking (22), and findings could have implications on future research, and the treatment with homeopathic interventions. Likewise, we believe rigid research is needed that compares generalized preventative prescribing, aside of the treatment of epidemics, to prescribing that is done following an actual appearance of symptoms. From such studies then, it could be identified which mode of prescribing has greatest value to homeopathic interventions and for patients above all.
The findings of this review suggest otherwise, but if such appraisal were to find generalized preventative prescribing to be efficacious it would provide a valuable treatment approach that could become an interesting tool in particular to professional athletes and sports persons. Within such generalized prophylactic homeopathic prescribing could lie the potential to enhance performance and reduce injury prevalence. Homeopathic remedies have little to no adverse effects (8,11) and, with few restrictions, are considered inept for doping efforts (Personal communication, 2012 June 19, 103). Should therefore, evidence be found, backing up the few experiential reports on performance enhancement with the use of homeopathic remedies (10, 104), this could extensively impact the future of sports. Until then, such reports deliver only anecdotal support for the use of homeopathic remedies as a stimulant to fitness for persons pursuing sports.
While the more conventional application of homeopathic remedies, for the treatment of injuries, sees its use already confirmed in the many successful treatments of the active lay athlete in every day practice, the news of such extensive use of homeopathy in professional sports as has been reported by the German Bundesliga clubs (11) is appreciated support, suggesting that effectiveness goes beyond research and is sufficiently documented by experience in practice.
Limits to our review
The trials and studies included in this review investigated diverse indications that were treated with different remedies or remedy complexes that were of differing dosage and potency, and were administered at differing frequency and via different routes of intake. The exercise inducing regimen also differed amongst the included studies. We considered this diversity of trial characteristics a limit to our investigation. We consequently judged it as difficult, if not inappropriate, to extract a consensus statement from trials of such differing attributes. Yet we do not believe that our early judgement of this induced bias to our review.
We had to draw a similar judgement as we proceeded to perform the review synthesis. The quality of conduction and reporting of trials differed largely and for a synthesising argument we needed to be able to identify a minimal amount of similarities. We therefore had to define further exclusion and inclusion criteria in order to be able to draw a synthesising conclusion from the findings. This was only possible for 4 of the included trials.
We believe our literature search was extensive, but have no assurance that we may have overseen or lacked to retrieve studies on this topic. We made no restrictions concerning the language in which studies were published and believe these issues to have minimal potential for bias in this review.
Conflicts of interest
The authors have no conflicts of interest to declare.
CONCLUSION
Much in line with the arguments of Walach (76) above, we had thought to undertake with this review an appraisal that combined the findings from two different types of research. As Downe (40) describes, such “Metasynthesis” aims to provide insight into discrepancies and aspect that are mutual in the findings in order to be able to extrapolate clarification on the approach reviewed. We have sought to do this for our investigation into the effectiveness of homeopathy for injuries from physical exertion, and have come to the conclusion that there is only inconsistent outcome to the debate of the effectiveness of homeopathic treatment for sports injuries.
Our review synthesis at best delivered some evidence for the effectiveness of the homeopathic complex preparation Traumeel and the single homeopathic remedy Arnica in the 4th decimal and 6th centesimal potency for sports related injuries, if they are applied topically to the affected area.
While our appraisal pointed out some of the advantages of homeopathy, it has also exposed the known gaps and problems within research, and has confirmed the aspects that are too often used to document the failings to eventually provide proof of homeopathic efficacy.
Our findings have nurture the debate over better quality trials and trial designs that are sensitive to factors influential in homeopathic treatment, and have confirmed that the research model used remains the greatest hurdle still and needs to be overcome in order to facilitate unbiased investigations into homeopathy.
The image of Homeopathy will not improve if we continue to produce research that lacks thorough and quality appraisal. There is no global consensus on research conduction and reporting, and as such accounts of trials differ greatly. An agreement on guidelines for trial investigations, such as the guidelines of the CONSORT statements, could provide a standardization that would assure quality investigations. Without consensus on what to report and how, internationally, there is no thorough evidence for the trustworthiness of a trialsˈ outcome.
For research into homeopathic interventions, the supplemental reporting guidelines to the CONSORT statement for RCT (37), the REDHOT guidelines (38), clarify only on matters pertaining to descriptive aspects of homeopathic relevance to the trial or study. Yet, these details fail to aid the reduction of bias due to flaws caused largely by the clash of homeopathic philosophies and the conventional research tools used in such an appraisal.
Such incongruity with homeopathic principles as has becomes apparent from our findings, and the discrepancies involved in research using the RCT as an investigating methodology, are factors that provoke fundamental falsification in investigations of homeopathic treatment effects. The flaws and biases noted in this review clearly show that neither the RCTˈs nor the observational studies are the ‘gold standard’ for investigations into holistic therapeutic interventions.
We argue that future research into homeopathic interventions should be sensitive to the diverse factors that potentially can flaw research at the very root. This we have mentioned in detail above, but we also recommend that future systematic appraisals and meta-analyses should pay attention to aspects that not only pertain to the research models used, but also pay attention to the diversity of indications, routes of administration and remedy potencies prescribed. We believe that a comparative analysis ignorant of these differences can only provide imprecise findings and inconsistent arguments. We are not aware of reviews that have in the past paid attention to such variations, but believe these factors to potentially, meaningfully impact outcomes.
We cannot conduct significant research with tools that do not respect the most fundamental dicta of a system of healthcare, and are ignorant of decisive aspects that are integral to realistic treatment situations. Only a methodology that does justice to a holistic treatment approach and delivers outcomes that are devoid of selection, reporting, funding and other bias, can finally deliver incontrovertible evidence of its effectiveness.
While we therefore propose a consensus on the reporting and conduction of trials in general, we further stress that for homeopathy it would be most favourable to have a research model that met criteria sensitive to the fundamental principles of this therapeutic approach. Therefore, a new methodology or a new synthesizing evaluation of trials using different trial methods is needed in order to be able to provide meaningful outcomes on the effectiveness of homeopathic treatments.
Unless the above mentioned frailties are accounted for, we will have to rely on good and not so good quality studies in reviews and for our informed decision making. Consequently we may fail to spot and to report of, the irrevocable scientific proof of the efficacy and the effectiveness of homeopathy. We have to make research into homeopathy fool-prove in order to inform our practice, and disperse critique.
Albeit the dissonant outcomes of the individual trials and studies, and the restrictive findings from the synthesis of this mixed systematic review, there remains some disbelief concerning the reported inconsistent outcomes. Where 97% of teams of a major football league resort to homeopathy for the treatment of their highly trained athletes, some evidence, be it reliant on the few positive scientific investigations, or of an experiential nature, must confirm the usefulness of the homeopathic preparations used. No organization, club or institution would expend on, and subject their stars to, a therapy or treatment that provided no positive impact.
Acknowledgements:
Thank you to Petter Viksveen for his translation of a study from Norwegian to English, and the Carstens Stiftung for the provision of some articles.
REFERENCES
1. Clarke, J. H. (2002). Homeopathy explained [Internet]. 2013 Jan 11 [cited 2013 January 11; Available from http://www.homeoint.org/books5/clarkehomeo/criticism.htm
2. De Schepper, L. Introduction: A Brief Overview of Hahnemann’s Life [Internet]. 1999 [cited 2013 January 11]; Available from http://www.wholehealthnow.com/homeopathy_pro/hahnemann_rev1.html
3. Ullman D. A Condensed History of Homeopathy [Internet]. 2013 January 1 [cited 2013 January 1]. Available from: http://www.homeopathic.com/Articles/Introduction_to_Homeopathy/A_Condensed_History_of_Homeopathy.html
4. Society of Homeopaths. Basic science [Internet]. 2013 [cited 2013 January 1]. Available from: http://www.homeopathy-soh.org/research/evidence-base-for-homeopathy-2/basic-science/
5. Hahnemann, S. Organon der Heilkunst (2.Auflage). Heidelberg: Karl.F.Haug Verlag; 1974.
6. Faculty of Homeopathy. The evidence base [Internet] 2013 [cited 2013 March 11]. Available from: http://www.facultyofhomeopathy.org/research/
7. Kisser R, Bauer R. The burden of sports injuries in the European Union. Research report D2h of the project “Safety in Sports”. Vienna: Austrian Road Safety Board (Kuratorium für Verkehrssicherheit); 2012.
8. Zittlau J. Blut an den Stollen [Internet]. 2008 [cited 2009 November 14] Available from: http://www.welt.de
9. Schmaranzer I. Homöopathie im Sport [Internet]. 2009 [cited 2010 March 20]. Available from: http://www.apoverlag.at
10. Kayne S. Homoeopathy in sports medicine. British Homoeopathic Journal. 1992; 81(3), 142-147. doi: 10.1016/S0007-0785(05)80560-5
11. Meyer D. EM-Profis setzen auf sanfte Medizin. Spiegel [Internet]. 2008 [cited 2009 August 13]; Available from http://www.spiegel.de
12. Naturheilmittel – Umfrage unter den Mannschaftsärzten der ersten und zweiten Fussballbundesliga. Wellnessing [Internet]. 2009 [cited 2009 November]; Available from: http://www.wellnessing.de
13. Subotnick S.Report from the medical practice – Treatment of Achilles Tendinopathy in the Athlete [Internet]. 2009 October 27 [cited 2009 October 27]. Available from: http://www.biopathica.co.uk
14. DZS. Individuell trainieren – Prof.Dr. P. Billigmann im Interview zum Thema Fussball [Internet]. 2010 March 20 [cited 2010 March 20]; Available from http://www.zeitschrift-sportmedizin.de
15. Professor Billigmann stärkt Sportler homöopathisch. Medical Tribune [Internet]. 2004 [cited 2009 November 14]; Available from http://www.medical-tribune.de
16. Kiewisz, N. (2009). Unkonventionelle Heilmethoden von Knie- und Sprunggelenkverletzungen bei Sportlern [Internet]. 2009 November 1 [cited 2009 November 1]; Available from http://archiv.ub.uni-heidelberg.de
17. Ernst, E. & Barnes, J. Are homeopathic remedies effective for delayed-onset muscle soreness? A systematic review of placebo controlled trials Perfusion. 1998; 11(1),4-8. doi: 10.1046/j.1365-2125.2002.01699.x
18. Ernst E, Pittler M. Efficacy of Homeopathic Arnica. Archives of Surgery. 1998; 133(11), 1187-1190. doi:10.1001/archsurg.133.11.1187
19. Morgan L. Homeopathic treatment of sports injuries. Healing Arts Press: Rochester, Vermont; 1988.
20. University of Victoria Libraries. What does peer-review mean? [Internet] 2011 [cited 2013 January 4]. Available from: http://library.uvic.ca/instruction/research/peerreview.html
21. Pschyrembel W. Pschyrembel Klinisches Wörterbuch (258. Auflage). Berlin: de Gruyter; 1998.
22. Mathie R, Hacke D, Clausen J, Nicolai T, Riley, D, Fischer P. Randomised controlled trials of homeopathy in humans: characterising the research journal literature for systematic review. Homeopathy 2013; 102, 3-24. doi: 10.1016/j.homp.2012.10.002.
23. CONSORT. Randomization [Internet]. 2007 [cited 2013 January 4]. Available from: http://www.consort-statement.org/resources/glossary/q—z/randomization/
24. Jepsen P, Johnsen S, Gillman M, Sørensen H. Interpretation of observational studies. Heart. 2004; 90(8), 956-960. doi: 10.1136/hrt.2003.017269
25. Einzelmittel, Komplexmittel [Internet] 2013 [cited 2013 April 11]. Available from: http://www.homoeopathie.de/komplexmittel.html
26. Raschka C, Trostel Y. Effekt einer homöopathischen Arnika-präparation (D4) auf Muskelkater. MMW-Fortschritte der Medizin Originalien. 2006; 148 (II), 93-95.
27. Research design service. Cross-over trials [Internet]. 2013 March 23 [cited 2013 March 23]. Available from: http://www.rds-sc.nihr.ac.uk/planning-a-study/study-design/quantitative-studies/clinical-trials/cross-over-trials/
28. Schmidt C. A double-blind, placebo-controlled trial: Arnica montana, applied topically to subcutaneous mechanical injuries. Journal of the American Institute of Homeopathy. 1996; 89(4), 186-193.
29. Schneider C, Schneider B, Hanisch J, van Haselen R. The role of a homeopathic preparation compared with conventional therapy in the treatment of injuries: An observational cohort study. Complementary Therapies in Medicine. 2008; 16, 22-27. doi: 10.1016/j.ctim.2007.04.004
30. Walker, B. The Anatomy of Sports Injuries. Berkeley, California: Lotus Publishing; 2007.
31. Cheung K, Hume P, Maxwell L. (2003). Delayed Onset Muscle Soreness. Sports Medicine. 2003; 33(2), 145-164. doi: 10.2165/00007256-200333020-00005
32. La Cava G. (1975). Definition and limits of the traumatology of sports*. Olympic Review [Internet]. 1975 [cited 2013 April 24]; (87-88),19-20. Available from http://www.la84foundation.org/OlympicInformationCenter/OlympicReview/1975/ore87/ore87f.pdf
33. Online Medical Information [Internet] 2013. Tendinopathy; [cited 2013 March 11]. Available from: http://online-medical-information.com/tendinopathy/tendinopathy.html
34. Quinn E. Tendonitis and Tendinopathy [Internet] 2011 [cited 2013 March 23]. Available from: http://sportsmedicine.about.com/od/paininjury1/a/tendonitis.htm
35. Lüdtke R, Wilkens J. Klinische Wirksamkeitsstudien zu Arnica in homöopathischen Zubereitungen [Internet]. 1998 [cited 2013 April 4]; Available from http://www.carstens-stiftung.de/wissen/hom/pdf/klin_wilkens_jb5.pdf
36. Vickers A. What conclusion should we draw from the data?. British Homeopathic Journal. 1995; 84(2), 95-101. doi: 10.1016/S0007-0785(95)80039-5
37. Schulz K, Altman D, Moher D. CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomised Trials. PLoS Med. 2010; 7(3). doi: 10.1371/journal.pmed.1000251
38. Dean M, Coulter M, Fischer P, Jobst K, Walach H. (2007). Reporting data on homeopathic treatments (RedHot): a supplement to CONSORT. Homeopathy. 2007; (96), 42-45. doi:10.10.16/j.homp.2006.11.006
39. von Elm E, Altman D, Egger M, Pocock S, Gøtzsche P, Vandenbroucke J. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. PLoS Med. 2007; 4(10). doi: 10.1371/journal.pmed.0040297
40. Downe, S. Metasynthesis: a guide to knitting smoke. Evidence Based Midwifery [Internet]. 2008 [cited 2012 Nov 22]; 6(1), 4-8. Available from http://www.doctoralmidwiferysociety.org/Portals/c8d3e3f8-9c01-4bf5-abd9-3fd6b4c510ae/Downe_S.pdf
41. Jawara N, Lewith G, Vickers A, Mullee M, Smith C. Homeopathic Arnica and Rhus toxicodendron for delayed onset muscle soreness. British Homoeopathic Journal. 1997; (86), 10-15. doi:10.1016/S0007-0785(97)80004-X
42. Plezbert J, Burke J. Effects of the homeopathic remedy Arnica on attenuating symptoms of exercise-induced muscle soreness. Journal of Chiropractic Medicine. 2004; 4(3), 152-161. doi: 10.1016/S0899-3467(07)60124-4
43. Tveiten D,Bruseth S,Borchgrevink C, Løhne K. Effekt av Arnika D30 ved hard fysisk anstrengelse.Tidsskrift for den Norske Laegeforening : Tidsskrift for Praktisk Medicin, ny Raekke. 1991; 111(30),3630-3631.
44. Tveiten D, Bruseth S, Borchgrevink C, Norseth J. Effects of the homeopathic remedy Arnica D30 on marathon runners: a randomized, double-blind study during the 1995 Oslo Marathon. Complementary Therapies in Medicine. 1998; 6, 71-74.
45. Vickers A, Fischer P, Smith C, Wyllie S,Lewith G. Homeopathy for delayed onset of muscle soreness: a randomised double blind placebo controlled trial. British Homeopathic Journal. 1997; 84(2), 95-101. doi: 10.1016/S0007-0785(95)80039-5
46. Vickers A, Fischer P, Smith C, Wyllie S, Rees R. Homeopathic Arnica 30x is ineffective for muscle soreness after long-distance running: A randomized, double-blind, placebo controlled trial. The clinical journal of pain [Internet]. 1998 [cited 2012 September 17]; 14(3), 227-231. Available from: http://ovidsp.tx.ovid.com/sp-3.8.1a/ovidweb.cgi?&S=PEMJFPNFEBDDCKAFNCOKPEDCNBOLAA00&Link+Set=S.sh.18%7c1%7csl_10
47. Cusmano L. Muscle Soreness – Do Sore Muscles Equal Muscle Building Results [Internet]. 2013 March 7 [cited 2013 March 7]; Available from http://www.shapefit.com/muscle-soreness-results.html
48. Birnesser H, Oberbaum M, Klein P, Weiser M. The homeopathic preparation TraumeelS compared with NSAIDs for symptomatic treatment of epicondylitis. Journal of Musculoskeletal Research 2004. 8(02n03), 119-128. doi: 10.1142/S0218957704001284
49. Schneider C, Klein P, Stolt P, Oberbaum M. A homeopathic ointment preparation compared with 1% Diclofenac gel for acute symptomatic treatment of tendinopathy. Explore. 2005; 1(6), 446-452. doi: 10.1016/j.explore.2005.08.010
50. Bemm T. Leistungssteigerung mit der Homöopathie. Pressetext.de [Internet]. 2009 [cited 2013 April 4]. Available from: http://www.aponeo.de/themenwelt/bewegung-und-sport/homoeopathie-im-sport/beitraege/leistungssteigerung-mit-der-homoeopathie/
51. Müller-Löbnitz C, Göthel D. Review of the clinical efficacy of the multicomponent combination medication Traumeel and its components. Alternative therapies [Internet]. 2011 [cited 2012 December 12]; 17(2), 18-31. Available from: http://www.biopathica.co.uk/Articles/Arthritis%20Rheumatic%20Conditions%20Sporting%20Injuries/81%20-%20Review%20of%20the%20Clinical%20Efficacy%20of%20Traumeel.pdf
52. Traumeel [Internet] 2013 [cited 2013 May 19]. Available from: http://www.heel.de/Heel-de-Search-Results.html?Keywords=Traumeel
53. Kaptchuk T. Powerful placebo: the dark side of the randomised controlled trial. The Lancet. 1998; 351(9117), 1722-1725. doi: 10.1016/S0140-6736(97)10111-8
54. Nuhn T, Lüdtke R, Geraedts M. Placebo effect sizes in homeopathic compared to conventional drugs – a systematic review of randomised controlled trials. Homeopathy. 2010; 99, 76-82. doi: 10.1016{j.homp.2009.11.002
55. Teixeira M, Guedes C, Barreto P, Martins M. The placebo effect and Homeopathy. Homeopathy. 2010; 99, 119-129. doi: 10.1016./j.homp.2010.02.001
56. Rogers P. Definition of eccentric weight training [Internet]. 2009 [cited 2013 April 11] Available from: http://weighttraining.about.com
57. Macmillan dictionary [Internet]. 2013. Prophylaxis; [cited 2013 March 23]. Available from: http://www.macmillandictionary.com/dictionary/british/prophylaxis
58. DocCheck Flexicon. [Internet]. 2013. Infiltration. [cited 2013 April 24]. Available from: http://flexikon.doccheck.com/de/Infiltration
59. Taylor W. Antidoting in homeopathic treatment [Internet]. 1998 [cited 2013 April 4]. Available from: http://www.vitalitylink.com/article-homeopathy-1085-antidoting-homeopathic-treatment-coffee-remedy-substance
60. Draper H, Wilson S, Flanagan S, Ives J. Offering payments, reimbursements and incentives to patients and family doctors to encourage participation in research. Family Practice. 2009; 26, 231-238. doi: 10.1093/fampra/cmp011
61. Product insert Traumeel S crème [Internet] 2009 [cited 2013 February 24]. Available from http://www.heel.com/upload/Traumeel_S_Tabl_K_014558_03_BPZ_7_1564.pdf
62. Product insert Voltaren Emulgel [Internet] 2005 [cited 2013 February 6]. Available from: http://www.parcelmed.de/pdf/3140374.pdf
63. Product insert Diclofenac-ratiopharm Gel [Internet] 2007 [cited 2013 February 6]. Available from: http://www.docmorris.de/medias/sys_master/8452192369044256.pdf?mime=application/pdf&realname=8510427.pdf
64. Statutes of the Research Council of Norway [Internet] 2011 [cited 2013 April 22]. Available from: http://www.forskningsradet.no/servlet/Satellite?blobcol=urldata&blobheader=application%2Fpdf&blobheadername1=Content-Disposition%3A&blobheadervalue1=+attachment%3B+filename%3D%22RCNStatutes2011.pdf%22&blobkey=id&blobtable=MungoBlobs&blobwhere=1274468227525&ssbinary=true
65. The Dove Healing Trust [Internet] 2012 [cited 2013 April 20]. Available from: http://opencharities.org/charities/800075
66. Blackie Foundation. The Blackie Foundation Trust – funding the future of homeopathy [Internet]. 2011 [cited 2013 April 22] Available from: http://www.blackieft.org/
67. History [Internet]. 2013 April 11 [cited 2013 April 11]. Available from: http://www.reckeweg.de/history.php?mid=47
68. Hevert – in the tradition of pastor Felke [Internet] 2013 [cited 2013 April 11]. Available from: http://www.hevert.com/en_US/company_profile/company_history
69. Mittelstadt U. Clever Homeopathy [Internet]. Uta Mittelstadt; 2011- To swallow or not to swallow…that is the question!; 2012 March 01 [cited 2013 April 24]. Available from: http://cleverhomeopathy.wordpress.com/2012/03/01/to-swallow-or-not-to-swallowthat-is-the-question/
70. Kleijnen J, Knipschild P, Riet G. Clinical trials of homeopathy. BMJ [Internet]. 1991 [cited 2012 December 17]; 302(6772), 316–323. Available from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1668980/
71. Society of Homeopaths. Research directory [Internet]. 2013 [cited 2013 April 21]. Available from: http://www.homeopathy-soh.org/research/how-to-find-out-more/research-directory/
72. Ernst E. Is homeopathy a clinically valuable approach? Trends in Pharmacological Sciences. 2005; 26 (11), 547-548. doi:10.1016/j.tips.2005.09.003
73. Linde, K., Clausius, N., Ramirez, G., Melchart, D., Eitel, F., Hedges, L. & Jonas, W. Are the clinical effects of homeopathy placebo effects? A meta-analysis of placebo-controlled trials. The Lancet. 1997; 350(9081), 834-843.
74. Concato J, Shah N, Horwitz R. Randomized, controlled trials, observational studies, and the hierarchy of research designs. The new England journal of medicine. 2000; 342,1887-1892. doi: 10.1056/NEJM200006223422507
75. Benson K, Hartz A. A comparison of observational studies and randomized, controlled trials. The new England journal of medicine. 2000; 342, 1878-1886. doi: 10.1056/NEJM200006223422506
76. Walach H, Falkenberg T, Fønnebø V, Lewith G,Jonas W. Circular instead of hierarchical: Methodological principles for the evaluation of complex interventions. BMC Medical Research Methodology. 2006; 6(29), 1-9. doi:10.1186/1471-2288-6-29
77. Golden I. Beyond Randomized controlled trials: Evidence in Complementary Medicine. Journal of evidence-based complementary & alternative medicine. 2012; 17(1), 72-75. doi: 10.1177/2156587211429351
78. Horn B, Balk J, Gold J. Revisiting the sham: Is it all smoke and mirrors?, Evidence-based complementary and alternative medicine. 2011; 4 pages. doi:10.1093/ecam/neq074
79. Corrigan P, Salzer M. The conflict between random assignment and treatment preference: implications for internal validity. Evaluation and program planning. 2003; 26(2), 109-121. doi: 10.1016/S0149-7189(03)00014-4
80. Walach H. The campaign against CAM and the notion of “evidence-based”. The Journal of alternative and complementary medicine. 2009; 15(10), 1139-1142. doi: 10.1089/acm.2009.0423
81. Relton C. Implications of the ˈplacebo effectˈ for CAM research. Complementary therapies in medicine. 2013; 21(2), 121-124. doi: 10.1016/j.ctim.2012.12.011
82. Walach H. Das Wirksamkeitsparadox in der Komplementärmedizin. Forschende Komplementärmedizin und klassische Naturheilkunde. 2001; 8, 193-195. doi:10.1159/000057221
83. Enck P, Klosterhalfen S. The placebo response in clinical trials – the current state of play. Complementary therapies in medicine. 2013; 21(2), 98-101. doi: 10.1016/j.ctim.2012.12.010
84. Kaptchuk T, Stason W, Davis R, Legedza A, Schnyer R, Kerr C et al. Sham device v inert pill: randomized controlled trial of two placebo treatments. BMJ. 2006; 332. doi: 10.1136/bmj.38726.603310.55
85. Flay B, Biglan A, Boruch R, Castro F, Gottfredson D, Kellam S et al. Standards of evidence: Criteria for efficacy, effectiveness and dissemination. Prevention Science. 2005; 6(3), 151-175. doi: 10.1007/s11121-005-5553-y
86. Walach H. The efficacy paradox in randomized controlled trials of CAM and elsewhere: Beware of the placebo trap. The Journal of Alternative and complementary medicine. 2001; 7(3), 213-218. doi:10.1089/107555301300328070
87. Walach H. The campaign against CAM – a reason to be proud. Journal of holistic healthcare [Internet]. 2009 [cited 2013 December 2012]; 6(1), 8-13. Available from: http://www.ecpm-europe.ch/Campaign_against_CAM.pdf
88. Shakya P, Madhav N, Shakya A, Singh K. Palatal mucosa as a route for systemic drug delivery: A review. Journal of controlled release. 2010; 151(1), 2-9. doi: 10.1016/j.jconrel.2010.11.003
89. Borchardt R. Value of pharmaceutical sciences. Journal of pharmaceutical and biomedical analysis 1998; 16(8),1387-1401. doi: 10.1016/S0731-7085(97)00165-9
90. Constantino H, Illum L, Brandt G, Johnson P, Quay S. Intranasal delivery: Physicochemical and therapeutic aspects. International journal of pharmaceutics. 2007; 337(1-2),1-24 doi: 10.1016/j.ijpharm.2007.03.025
91. Chin R, Lee B. Principles and practice of clinical trial medicine. London: Elsevier Inc; 2008.
92. Carlston, M. Classical Homeopathy. Pennsylvania: Churchill Livingstone; 2003.
93. Clarke, J. The Prescriber. Somerset: Hillman Printers (Frome) Ltd; 1992.
94. Kayne S. Homeopathic Pharmacy: An Introduction and Handbook. New York: Churchill Livingstone; 1997.
95. Pratt N. Homeopathic Prescribing. Beaconsfield: Beaconsfield Publishers Ltd; 1991.
96. Schütt K. Application of Homeopathic Remedies. Hpathy [Internet]. 2009 [cited 2011 February 16]. Available from: http://hpathy.com/homeopathy-papers/application-of-homeopathic-remedies/
97. Shikiar R, Rentz A. Satisfaction with medication: An overview of conceptual, methodologic and regulatory issues. Value in health. 2004; 7(2), 204-215. doi: 10.1111/j.1524-4733.2004.72252.x
98. Homeopathic prophylaxis: Fact or fiction [Internet] 2006 [cited 2013 March 23]. Available from: http://nationalcentreforhomeopathy.org
99. Bracho G, Varela E, Fernández R, Ordaz B, Marzoa N, Menéndez J et al. Large-scale application of highly-diluted bacteria for Leptospirosis epidemic control. Homeopathy 2010; 99(3), 156-166. doi: 10.1016/j.homp.2010.05.009
100. What Is A Pandemic? What Is An Epidemic? [Internet] 2009 [cited 2013 February 12]. Available from: http://www.medicalnewstoday.com
101. What Are Epidemics, Pandemics, and Outbreaks? [Internet] 2013 [cited 2013 February 12]. Available from: http://www.webmd.com
102. Maes J, Kravitz L. Treating and Preventing DOMS [Internet]. 2003 [cited 2013 February 4]; Available from http://www.ideafit.com
103. What about homeopathic products and alternative medicine? [Internet] 2013 [cited 2013 February 2]. Available from: http://www.wada-ama.org/en/Resources/Q-and-A/Athletes-and-Medications/
104. Barrois D. Entraînement du sportif en homéopathie. Homéopathie française.1998; 76(6), 351-318.

